| Posted: Oct 18, 2016 |
'Bolt of lightning' captures development of block copolymer
(Nanowerk News) The ability to precisely control every aspect of a material, even at the nanoscale, is of critical importance in a host of applications.
|
|
One class of such novel materials, block copolymers (BCPs), are being developed today to enable continued advancement of data archiving, as well as advanced drug refinement using protein filters, among other things.
|
|
BCPs are combinations of materials that, like oil and water, don’t readily mix in nature but can be forced to do so through chemical “marriage” of blocks in a polymer chain. A group of Cornell engineers led by Mike Thompson, M.A. ’82, Ph.D. ’84, and Chris Ober, professors of materials science and engineering, are exploring how to control these novel materials by capturing the early stages of structure development using laser induced ultra-fast heating and cooling.
|
|
Their paper was published in the American Chemical Society journal, Macromolecules ("Kinetics of Block Copolymer Phase Segregation During Sub-millisecond Transient Thermal Annealing"). Alan Jacobs, a graduate student in Thompson’s lab, is lead author.
|
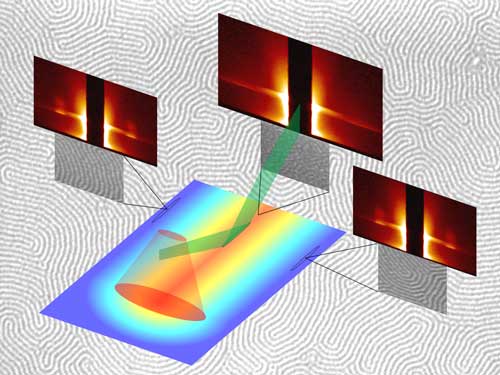 |
| A schematic representation of the experimental and measurement setup for block copolymer phase segregation. (Image: Alan Jacobs)
|
|
Thompson said that heating these materials for extremely short durations – between 250 microseconds and 10 milliseconds – allows researchers to work with temperatures in excess of 1,000 degrees even in thermally sensitive polymer materials.
|
|
“This opens up a whole new regime for studying the dynamics of polymer motion,” he said. “Normally we think of polymers as being fairly rigid. But when you go to high enough temperatures, they become exceedingly mobile and flexible in a liquid-like state. With laser induced heating, we can reach these temperatures fast enough, and also cool down fast enough, that the block copolymers can reorder and restructure themselves into useful and interesting structures before they begin to burn.”
|
|
BCPs order themselves based on the chemistry of their constituent “blocks,” commonly referred to as A’s and B’s. As the two blocks do not want to mix, these material try to find structures where mixing is minimized.
|
|
“If you have approximately the same amount of the A and B materials,” Thompson said, “they form layers of A and B. … All the A blocks try to get next to each other, as do all the B blocks.”
|
|
But by rapid heating and cooling, these blocks that normally wouldn’t get along are forced together by thermal motion, generating unique materials and patterns.
|
|
“With enough energy in there,” Jacobs said, “they will mix randomly and create a more homogeneous structure at high temperature. Then, as they cool, they are free to adopt the new and unique structures.”
|
|
In work performed at the Cornell NanoScale Science and Technology Facility and the Cornell Center for Materials Research and analyzed at the Cornell High Energy Synchrotron Source, the group heated a BCP built from polystyrene (PS) and polymethyl methacrylate (PMMA), using a laser spike annealing apparatus to temperatures up to 550 C (more than 1,000 F) for durations no longer than 10 milliseconds.
|
|
The material did not degrade during that extremely short anneal time, but instead was put in a state of controlled disorder, which could be tuned by varying both the duration of the heating and the peak temperature. Although perhaps counterintuitive, this controlled disorder results in a final material that has fewer defects and unique properties, which can be “designed” by both the underlying chemistry and the annealing.
|
|
The group is, in essence, capturing a moment in time by hitting the BCP with a “bolt of lightning.” Future work is focused on understanding the details of the process. “We’ve shown that we can get fewer defects using this really rapid heating to initially do phase segregation, but we don’t yet know why,” Jacobs said. “But this gives us a good start, a good idea of what’s going on in those initial stages.”
|

