| Posted: Nov 08, 2016 |
Fast and easy two-step creates new porous materials
(Nanowerk News) Materials scientists at Pacific Northwest National Laboratory have come up with a new approach to synthesizing porous materials leading to the creation of a new class of materials called coordination covalent frameworks (CCFs). This approach uses the combination of MOF and COF chemistry in two steps, making the synthesis process faster and easier. Their results appear in ACS Applied Materials and Interfaces ("Coordination Coordination Frameworks: A New Route for Synthesis and Expansion of Functional Porous Materials").
|
|
Dr. Praveen Thallapally, lead author of the paper, used a popular building toy to illustrate the process. “Much like Tinkertoys®, which are sticks and circular pieces with holes drilled in them used to create structures, we substitute metal ions for the circular pieces and organic molecules for the sticks to create MOFs,” he said, “whereas in COFs, the circular pieces were replaced by covalent bonds and sticks with organic molecules. For CCFs we use the MOF chemistry in the first step and COF chemistry in the second step. The result is a CCF with a large surface area.”
|
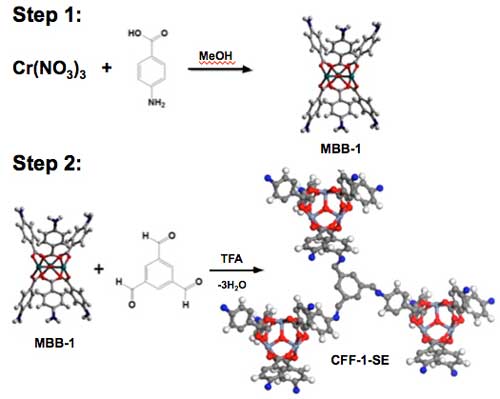 |
| Illustration of the two-step synthesis approach. Step 1 shows MOF chemistry to generate isolated MBB-1, and Step 2 shows the polymerization reaction of MBB-1 with tri-aldehydes (COF chemistry) to result in a CCF-1.
|
|
According to Thallapally, Dr. Sameh Elsaidi, a postdoctoral researcher in the materials group, came to him one day with a new view of the Tinkertoy model.
|
|
“Sameh had created a non-porous (no surface area) molecular building block (MBB) in one step with metal clusters (circular pieces) and organic linker (sticks) that has an amino group at one end of the linkers. This became the basis of the new, two-step approach that resulted in CCFs,” Thallapally said. The condensation reactions were used in COFs between amines and aldehydes to form an extended structure. The same COF approach was used in the second step to create a CCF material.
|
|
The MBBs were isolated using 4-aminobenazoic acid and Cr(III) salt. They were then polymerized, a process that forms strong covalent bonds where small organic molecules can connect the MBBs, forming extended porous CCF material with high surface area that can selectively separate gases.
|
|
Why It Matters: The researchers have basically eliminated the multi-step synthesis to make extended organic linkers that were typically used in the MOF and COF synthesis to form a high-surface-area material. In principle, the two-step method can create high-surface-area materials.
|
|
The scientists used Fourier transform infrared analysis to prove the formation of amide or imine bonds in the CCF structures and show how this material is different than the original MOF building block. Gas adsorption measurement revealed that all polymerized CCFs were permanently porous. Thus the two-step approach opens a new avenue to this class of materials that merge coordination (MOF) and covalent synthesis (COF) approaches.
|
|
What’s Next: The scientists hope to adapt the two-step approach to create CCFs with different catalytic sites within the framework. They hope to patent the process and materials.
|

