| Posted: Dec 07, 2016 |
Uncovering the secrets of water and ice as materials
(Nanowerk News) Water is vital to life on Earth and its importance simply can’t be overstated -- it’s also deeply rooted within our conscience that there’s something extremely special about it. Yet, from a scientific point of view, much remains unknown about water and its many solid phases, which display a plethora of unusual properties and so-called anomalies that, while central to water’s chemical and biological importance, are often viewed as controversial.
|
|
This inspired researchers at University College London and Oxford University to pursue a better understanding of water and ice as materials, which has a far-reaching impact on many areas of research. In an article in The Journal of Chemical Physics ("Detailed crystallographic analysis of the ice VI to ice XV hydrogen ordering phase transition"), they report their work on the hydrogen ordering of the disordered ice VI phase relative to its ordered counterpart ice XV.
|
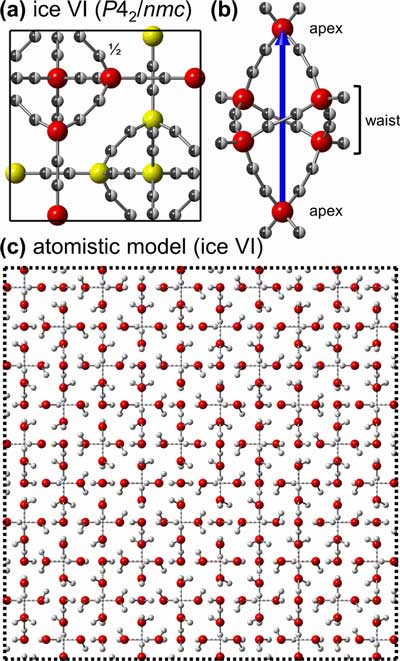 |
| (a) Unit-cell projection of ice VI representing the average P42/nmc structure (origin choice 2) viewed along the [001] direction. The oxygen atoms of the two different networks are shown in red and yellow, respectively. The hydrogen positions are half occupied. (b) The hexameric unit of a single network viewed along [110]. The four-fold rotoinversion axis is shown as a blue vector. (c) Atomistic model of a 5 × 5 × 6 supercell consistent with fully hydrogen-disordered ice VI. The maximal deviation from ½ occupancy is 0.013 with an average deviation of 0.005. For clarity, only atoms in the 0 < z < 1/6 range are shown. Covalent O–H bonds are indicated by solid lines, and O···H hydrogen bonds by dashed lines. (© AIP)
|
|
“Whenever liquid water freezes, only its oxygen atoms actually end up in fixed positions,” explained Christoph G. Salzmann, associate professor and Royal Society Research Fellow, Department of Chemistry, University College London. “The hydrogen atoms remain disordered -- so we call such phases of ice ‘hydrogen disordered.’ Upon cooling, the hydrogen atoms are expected to become ordered and result in hydrogen-ordered ices. Yet, this process is difficult because the reorientations of the hydrogen-bonded water molecules are highly cooperative.”
|
|
To help explain the concept, he used a tile game as an analogy.
|
|
“Moving from disorder to order is difficult work because the tiles can’t move independently -- similar to the situation in ice,” he said. “But, a few years ago, we found that adding a small amount of hydrochloric acid dramatically helps achieve hydrogen order at low temperatures.”
|
|
Hydrochloric acid is the “magic ingredient” that speeds up the reorientations of the water molecules.
|
|
Ice VI and ice XV are both high-pressure phases of ice that form at about 10,000 atmospheres. “The structure of ice XV has been the topic of lively scientific discussion for years,” Salzmann said. “A variety of different and, in part, conflicting models have been suggested from both experimental data -- including a previous study by our group -- as well as computational studies.”
|
|
For this work, the researchers turned to neutron diffraction to analyze the structure of ice XV and its formation from ice VI. “Using neutrons is important because X-rays are essentially ‘blind’ toward hydrogen atoms,” Salzmann said. “To fully solve the structure of ice XV, we really need to know where the hydrogen atoms are located -- neutrons are essential.”
|
|
The group’s work represents a major change in the understanding of ice XV that consolidates much of their previous work. “First, we’ve shown using neutron diffraction at the ISIS Science and Technology Facilities Council in the U.K. that the ice shrinks in two directions, but expands in the third during the transition from ice VI to XV,” he explained. “Using density functional theory calculations, we can show that only one particular structural model of ice XV is consistent with these changes.”
|
|
Incidentally, this structure is also the one the group proposed from their in-depth analysis of neutron data.
|
|
“This agreement between experiment and calculations is great, in particular, because there have been conflicting views regarding ice XV,” he added. “The overall volume of the ice increases during the phase transition, which finally explains why the transition is observed more readily at ambient pressure than at higher pressures -- behavior that has puzzled us for a long time.”
|
|
Another key point, presented in their article, is a new computer program called “RandomIce,” which has produced the best structural description of ice XV to date. “We’ve presented the preparation of the most ordered ice XV to date, but we haven’t achieved a completely ordered state,” Salzmann said.
|
|
RandomIce enables preparing large-scale molecular models, which the group calls “supercells.” These are consistent with the average structure obtained from the diffraction data, and essentially RandomIce is “playing” the tile game described earlier until the best agreement with the diffraction data is achieved. “To do this, more than 100 million ‘tile moves’ were necessary,” Salzmann pointed out.
|
|
The group’s work opens the door to the development of more accurate computer models of water that can benefit a wide range of disciplines -- from biology and chemistry to geology and the atmospheric sciences.
|
|
Further, it’s now possible to “clarify in which form ice is expected to occur under certain pressure and temperature conditions inside icy moons and planets,” Salzmann said.
|
|
What’s next for the researchers?
|
|
“There’s still an open question about why we can’t achieve full order in ice XV,” Salzmann said. “We’ve already started new experimental work to explore how the properties of ice change within nanoconfinements and the presence of chemical species -- because we’re interested in understanding the complex behavior of ice on comets and within our atmosphere.”
|

