| Posted: Mar 31, 2017 |
Graphene lid revitalizes imaging technique
(Nanowerk News) By capping liquids with graphene, an ultrathin sheet of pure carbon, researchers at the National Institute of Standards and Technology (NIST) and their colleagues have revitalized and extended a powerful technique to image surfaces. The graphene lids enable researchers for the first time to easily and inexpensively image and analyze liquid interfaces and the surface of nanometer-scale objects immersed in liquids. The new capability has the potential to advance the development of batteries, highly charged capacitors for power-grid technology, and new catalysts such as those used in the chemical industry.
|
|
In the imaging technique, known as photoemission electron microscopy (PEEM), ultraviolet light or X-rays bombard a sample, stimulating the material to release electrons from a region at or just beneath its surface. Electric fields act as lenses, focusing the emitted electrons to create an image.
|
 |
| Experimental set-up shows an array of graphene-capped liquids. The caps enable the liquids to be studied using an image technique that previously was restricted to studying solid surfaces. (Image: A. Kolmakov/NIST)
|
|
Researchers have used the method for decades to discern such fine-scale features as the patterns of chemical reactions on the surface of catalysts, as well as the magnetic field structure of memory devices and the molecular architecture of biological compounds. But PEEM has typically been restricted to solid surfaces that are in a high-vacuum environment. The method hasn’t had the ability to study liquids and gases at ordinary pressures. A liquid sample, for instance, would evaporate and create sparks if directly exposed to the high vacuum in the PEEM setup.
|
|
In the past, scientists have attempted to overcome these challenges by using a technique known as differential pumping, which bridges the gap between the high pressure of the sample and the essentially zero pressure of the microscope. But such instrumentation is not sufficient to reach truly ambient pressure conditions and is too expensive and not widely accessible for routine use, notes NIST physicist Andrei Kolmakov.
|
|
Instead, he and his colleagues from NIST, the University of Maryland, the University of Saskatchewan, the Canadian Light Source and Oregon State University developed a cost-effective and easy-to-implement alternative. Sealing a liquid or gaseous sample with a graphene lid just one or two atomic layers in thickness keeps the sample at atmospheric pressure while allowing the system to be placed under vacuum.
|
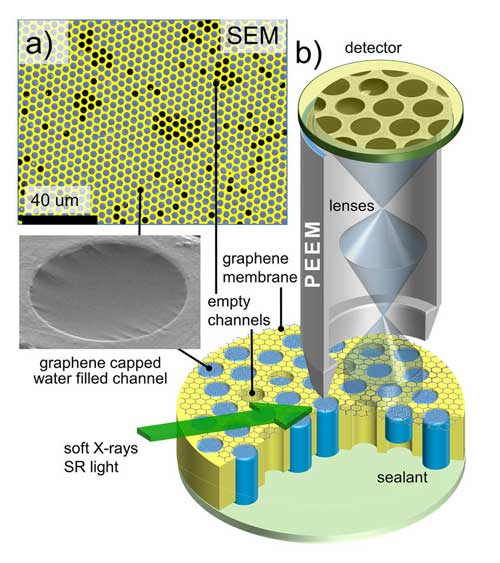 |
| X-rays illuminate a graphene-capped array of liquids, prompting molecules in solution to emit electrons. The graphene caps allow the electrons to pass freely, carrying information on the chemical state of the molecules, but prevent water from leaking out, ensuring that the liquid samples do not dry out. (Image: A. Strelkov /NIST)
|
|
In a recent issue of Nano Letters ("Enabling Photoemission Electron Microscopy in Liquids via Graphene-Capped Microchannel Arrays"), the scientists reported that the graphene lid enabled electrons emitted by the test liquid to pass nearly unimpeded to the detector, yet kept the liquid from escaping into the vacuum of the PEEM. An array of the lids retained the liquid samples for hours under high vacuum, long enough to perform routine electron imaging and spectroscopy experiments.
|
|
“This very simple solution, adding a layer of graphene,” allows researchers to use PEEM in its standard configuration without the need for extra and expensive equipment, said Kolmakov.
|
|
“The concept of extending the use of PEEM to probe liquids is in a way revolutionary,” commented physicist Andrea Locatelli of Synchrotron Radiation Laboratory Elettra in Trieste, Italy, who was not part of the research team. “Countless applications can indeed be foreseen,” he added.
|
|
For instance, noted Kolmakov, the lids allow the liquid to be changed while an experiment is in progress, helping researchers to understand the behavior of the sample under different chemical environments. In addition, because the setup uses an array of identical lids, each can be a different sample, and the technique can be used in conjunction with powerful statistical analysis, data mining and pattern recognition methods.
|


