| Posted: May 30, 2017 |
Tricking molecules into creating new nano-shapes
(Nanowerk News) Making small structures, at the nanometer length scale, is extremely difficult. However, these structures are vital for faster, denser more energy-efficient computing devices.
|
|
Scientists have devised materials that can create complex three-dimensional structures. They did it by exploiting how molecules self-assemble, spontaneously packing into nano-sized shapes.
|
|
Also, they took advantage of how materials respond to the surfaces they are cast on. When correctly designed, materials can be coaxed into never-before-seen structures.
|
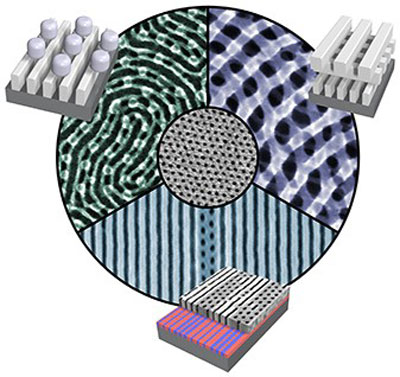 |
| Diverse, never-before seen nanostructures. Scientists developed a suite of methods that can coax simple nanomaterials (example shown in center) into spontaneously forming much more complex nanostructures. Three examples are shown, along with schematics of the three-dimensional order. (Image: Center for Functional Nanomaterials, Brookhaven National Laboratory)
|
|
This work greatly broadens the diversity and complexity of nanostructures that can be formed using self-assembling molecules. Today’s self-assembling materials make simple repeating patterns. The new method allows for complex structures. These structures mean forming different nano-patterns in different parts of a material. More complex structures will enable better microchips, water filter membranes and batteries.
|
|
Self-assembly is a powerful concept for making nanoscale structures where molecules are designed to spontaneously pack themselves into the desired shape or pattern without lithographic patterning.
|
|
However, conventional self-assembly yields a small library of very simple shapes. In this work, scientists at the Center for Functional Nanomaterials (CFN) devised responsive self-assembling systems, where the molecules reorganize in response to external influences.
|
|
In particular, this enables simple molecules to form much more complex patterns—indeed they can form three-dimensional nanostructures that would never form using conventional self-assembling materials.
|
|
Scientists at the CFN used thin films of block copolymers (BCP)—chains of two distinct molecules linked together. Through well-established techniques, the scientists spread BCP films across a substrate, applied heat, and watched the material self-assemble into a prescribed configuration. To make subsequent layers “talk to each other,” the team infused each layer with a vapor of inorganic molecules to seal the structure, which allowed each layer to act as a template for the one above it.
|
|
This technique demonstrated the formation of a broad range of nanostructures never before observed. This fundamental breakthrough substantially broadens the diversity and complexity of structures that can be made with self-assembly, and correspondingly broadens the range of potential applications. For example, intricate three-dimensional nanostructures could yield transformative improvements in nano-porous membranes for water purification, bio-sensing, catalysis, or dense computer memories and microprocessors.
|
Publications
|
|
A. Stein, G. Wright, K.G. Yager, G.S. Doerk, and C.T. Black "Selective directed self-assembly of coexisting morphologies using block copolymer blendsExternal link." Nature Communications 7, 12366 (2016). [DOI: 10.1038/ncomms12366]
|
|
A. Rahman, P.W. Majewski, G. Doerk, C.T. Black, and K.G. Yager. “Non-native three-dimensional block copolymer morphologiesExternal link.” Nature Communications 7, 13988 (2016). [DOI: 10.1038/ncomms13988]
|

