| Posted: Jun 14, 2017 |
Making hydrogen fuel from humid air
(Nanowerk News) One of the biggest hurdles to the widespread use of hydrogen fuel is making hydrogen efficiently and cleanly. Now researchers report in the journal ACS Nano ("Surface Water Dependent Properties of Sulfur-Rich Molybdenum Sulfides: Electrolyteless Gas Phase Water Splitting") a new way to do just that.
|
|
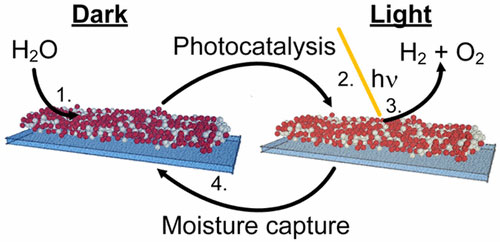
They incorporated a photocatalyst in a moisture-absorbing, semiconducting paint that can produce hydrogen from water in the air when exposed to sunlight. The development could enable hydrogen fuel production in almost any location.
|
 |
|
Traditionally, hydrogen destined for industrial use has come from fossil fuels. But this approach creates carbon byproducts and other pollutants.
|
|
In search of a cleaner source, researchers have turned to water as a source of hydrogen. Current methods to split water focus on its liquid form and thus require liquid electrolytes, which lead to high cost, inefficiency and other technical challenges.
|
|
These drawbacks could be overcome by using water in its gas phase, but few studies have explored this strategy. So Torben Daeneke, Kourosh Kalantar-zadeh and colleagues set out to fill this void.
|
|
Using a simple, scalable method, the researchers developed a photocatalyst to generate hydrogen from water vapor using a highly porous, sulfur-rich molybdenum sulfide. The compound belongs to a class of highly conductive materials previously recognized as efficient water-splitting catalysts in liquid.
|
|
Testing showed that the sulfide strongly absorbed moisture from the air. Then, combining the sulfide with titanium dioxide nanoparticles, the researchers created an ink that can be coated onto surfaces, such as glass. Films printed with the ink produced hydrogen without electrolytes or external power sources at a relatively high rate.
|
|
The moisture-absorbing photocatalytic paint can be applied to any surface such as building facades, introducing the novel capability of generating hydrogen fuel just about anywhere.
|

