| Posted: Jul 26, 2017 |
Getting closer to porous, light-responsive materials
(Nanowerk News) Researchers at Kyoto University's Institute for Integrated Cell-Material Sciences (iCeMS) and the University of Tokyo have developed a light-responsive crystalline material that overcomes challenges faced in previous studies.
|
|
Photochromic molecules change their electronic states or chemical structures, when exposed to light. They can play key roles in the development of 'photoresponsive' materials that could be used in delivery systems for controlled drug release, or to develop dynamic scaffolds for tissue engineering, among other applications. But so far, their use with solid materials has proven challenging because the materials have been too rigid to allow repeatable and reversible changes.
|
|
Susumu Kitagawa of iCeMS, Hiroshi Sato of the University of Tokyo, and their colleagues, prepared a flexible porous crystal composed of a photoresponsive dithienylethene derivative, zinc ions (Zn2+), and 1,4-benenzenedicarboxylate.
|
|
The 'porous coordination polymer' consisted of two-dimensional sheets connected by pillars of photoresponsive molecules, which created a three-dimensional, entangled framework. The researchers compare the entwined components to twisted metal wire and string puzzles.
|
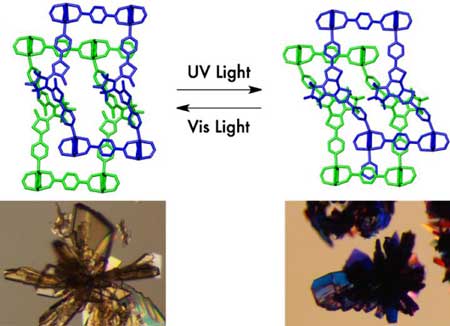 |
| Entangled porous coordination polymers like 'wire-and string puzzles' enable reversible and repeatable photomodulation of CO2 sorption. The single crystals of the porous material showed a drastic color change upon irradiation of ultraviolet and visible light. (Image: Kyoto University iCeMS)
|
|
Due to the flexible nature of the entangled framework, the channels changed shape when exposed to light. The distance between the two layers shrank upon ultraviolet irradiation then expanded when lit by visible light.
|
|
The researchers tested the material's ability to uptake carbon dioxide (CO2). When the material was not irradiated, it adsorbed up to 136 millilitres (ml) of CO2. When exposed to ultraviolet light, the pores shrank, decreasing CO2 adsorption to 108 ml. When then exposed to visible light, CO2 absorption rose again to 129 ml. It then decreased to 96 ml upon re-exposure to ultraviolet light.
|
|
The polymer's entangled framework enables these reversible and repeatable CO2 absorption changes; it gives room for the photoresponsive molecules to transform while allowing them to release their strain into the flexible material.
|
|
Preliminary tests indicated that the porous crystal could also adsorb other gases, such as nitrogen, at various temperatures, but more detailed investigation is required.
|
|
"Our strategy will grant access to a new dimension of porous compounds as platforms for various photochemical conversions and the photomodulation of porous properties," conclude the researchers in their study, published in the journal Nature Communications ("Flexible interlocked porous frameworks allow quantitative photoisomerization in a crystalline solid").
|

