| Posted: Aug 30, 2017 |
Two for the price of one: Exceeding 100 percent efficiency in solar fuel production
(Nanowerk News) In conventional systems for converting sunlight to chemical fuels, a single photon, no matter how energetic, leads to a single electron-hole pair and thus a single electron-hole pair’s worth of chemical work. The one-to-one situation limits devices that use sunlight to create chemical fuels.
|
|
Scientists have overcome this limit. They demonstrated for the first time that two electron-hole pairs, and two electron-hole pair’s worth of chemical work, can be produced from a single photon (Nature Energy, "Multiple exciton generation for photoelectrochemical hydrogen evolution reactions with quantum yields exceeding 100%" and Science, "Peak External Photocurrent Quantum Efficiency Exceeding 100% via MEG in a Quantum Dot Solar Cell").
|
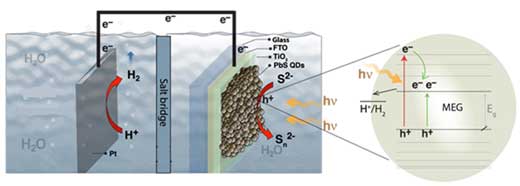 |
| When lead sulfide (PbS) quantum dots (QDs) absorb high-energy photons (right), two electron-hole pairs can be produced. Both electron-hole pairs can participate in chemical reactions (left) to produce hydrogen fuel.(© NPG) (click on image to enlarge)
|
|
This research could make the conversion of sunlight into chemical fuels more efficient by harvesting more energy from highly energetic photons. Conventional systems discard this excess energy as heat. Higher efficiency, in turn, means lower cost. Why? It lets the device produce more fuel per unit area of the solar cell and of the land supporting the cell.
|
|
In conventional semiconductor-based energy harvesting of photons from the sun, excess photon energy above the semiconductor bandgap is wasted. Recently, scientists showed that semiconductor quantum dots could enable harvesting of the excess photon energy through multiple exciton generation: greater than 100 percent external quantum efficiency, a signature of multiple exciton generation, for producing electricity from light was demonstrated in a solar photovoltaic cell.
|
|
In this work, scientists demonstrated greater than 100 percent external quantum efficiency in a solar photoelectrochemical cell, the first example of enhanced yield of a chemical reaction by multiple exciton generation.
|
|
The photoelectrochemical cell consisted of a lead sulfide (PbS) quantum dot photoelectrode deposited from solution using a layer-by-layer approach on top of a patterned sol-gel TiO2/FTO (F-doped SnO2) coated glass slide.
|
|
The team demonstrated hydrogen evolution from aqueous Na2S solutions with external quantum efficiencies greater than 100 percent for quantum dot photoelectrodes with band gap energies of 0.85, 0.92 and 1.08 eV, and for incident photon energies greater than 2.7 times the bandgap energy.
|

