| Posted: Oct 06, 2017 |
What can be discovered at the junction of physics and chemistry?
(Nanowerk News) TSU scientist Rashid Valiev and colleagues from the universities of Helsinki and Oslo have discovered a new type of rare molecules whose properties can be controlled by changing the induction of an external magnetic field. These are paramagnetic molecules from the class porphyrins. Porphyrins are part of hemoglobin and chlorophyll and are closely related to the processes of photosynthesis and respiration in living organisms.
|
|
The results of the study were published in the journal Chemical Communications ("Closed-shell paramagnetic porphyrinoids").
|
 |
| These are closed-shell paramagnetic porphyrinoids. (Image: Rashid Valiev)
|
|
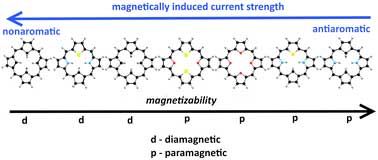
Open paramagnetic porphyrins with a closed electron shell are very rare molecules, because they have a specific electronic structure. Usually, molecules with such a structure are very unstable, and open porphyrins, on the contrary, are unchanged even in the air around us. This makes it possible to manipulate their physicochemical properties with an external magnetic field in various applied fields of magnetooptics and nanotechnology.
|
|
Since 2012, a group of scientists, which includes an assistant professor at TSU, has studied the aromatic nature of porphyrins and their derivatives. Aromaticity is a special property of some chemical compounds to exhibit anomalously high stability. That is an important concept in theoretical chemistry and is closely related to the problem of classifying and arranging organic molecules according to their reactivity. However, scientists define it using physics, in particular, they calculate in the molecules magnetically induced currents.
|
|
"For me, it was always interesting for the physicist to connect our currents and the concept of aromaticity with the spectroscopic or physical properties of molecules," says Rashid Valiev. "This was done in 2017 for highly antiaromatic porphyrins. Such molecules can be used in magnetooptical problems, where the control of physical properties of molecules is used by changing the induction of an external magnetic field. The fundamental significance of our result is that we explained the nature of the paramagnetism of these molecules."
|
|
Using theoretical methods of quantum chemistry, Valiev and his colleagues from the universities of Helsinki and Oslo studied the magnetic properties of seven molecules of isoflorines and carbaporphyrins, both synthesized and hypothetical. They showed that four of the seven molecules considered exhibit paramagnetism, and their spin is zero in the ground electronic state, which is an extremely rare case.
|

