| Posted: Feb 01, 2018 |
Splashdown: Supersonic cold metal bonding in 3-D
(Nanowerk News) When a fragile surface requires a rock-hard, super-thin bonded metal coating, conventional manufacturing processes come up short. However, Cold Gas Dynamic Spray (CGDS) can do just that - with a big caveat. CGDS is enormously versatile, but is also very difficult to predict key aspects of the entire process.
|
|
Now a temperature-based 3D model by Professor Tien-Chien Jen from the University of Johannesburg starts unlocking the mysteries of the CGDS film-growing process in the particle deposition zone (Journal of Thermal Spray Technology, "Particle Bonding Mechanism in Cold Gas Dynamic Spray: A Three-Dimensional Approach").
|
|
The model is the first to connect the dots between particle impact velocity, energy transformation, and temperature rise in the particle impact zone, in three dimensions.
|
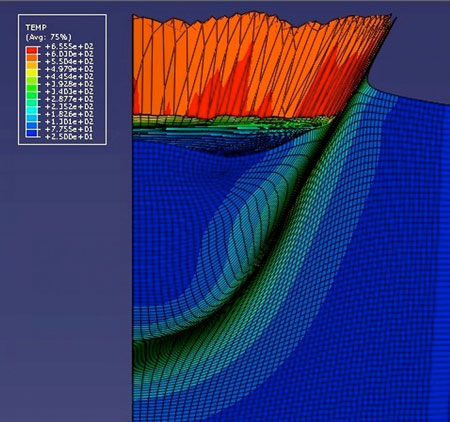 |
| The Cold Gas Dynamic Spray (CGDS) process is already used in the manufacture of aircraft parts and other delicate surfaces. However the entire CGDS process from flight-zone to particle deposition zone is very hard to predict numerically. The CGDS deposition zone is now 3-D modeled to show the effects of temperature for the first time, by Professor Tien-Chien Jen from the University of Johannesburg. In this image, the model predicts an orange/red "splash" of aluminum substrate just after a 5 micron copper particle has impacted it at 700 meters per second. (Image: Hong-Shen Chen, University of Johannesburg) (click on image to enlarge)
|
|
CGDS is already used extensively to manufacture or repair metal parts for large passenger airliners, as well as mobile technology and military equipment.
|
|
In the process, a de Laval nozzle sprays micron-sized metal particles over a short distance, typically 25mm, at a metal or polymer surface. The particles impact the surface at speeds ranging from 300 meters per second to 800 meters per second. As a frame of reference, the speed of sound is 343 meters per second.
|
|
CGDS has the best temperature range of all industrial spraying processes in use today, and saves energy, because no heating is added.
|
Manufacturing mystery
|
|
A mystery starts on the factory floor, however. If you have a 5 micron copper particle, how fast should it arrive at the deposition zone on aluminum, if you haven't used this combination before? Or you select a new metal for the particles, and a new metal for the surface: how do you even start guessing what size the particles should be, and at what speed they should impact the surface to give you a well-bonded coating?
|
|
Mind, the CGDS bonded coating should come without evaporation, crystallization, residual stresses or other thermal damages - some of the big reasons CGDS is used in the first place. These questions can have enormous financial implications for the manufacturing machines on the factory floor.
|
Physics still out
|
|
Why CGDS bonds metal particles to a substrate surface has defied understanding since its invention in the 1980s by the military, says Jen, Professor in the Department of Mechanical Engineering Science at the University of Johannesburg.
|
|
"At first, the military used CGDS to repair spare parts in the middle of nowhere. Then other industries realized you can use it on very fragile surfaces as well. You can create a new bonded surface only a few microns thick, or keep spraying until you have a 10mm coating. Once you've filled in the cracks or holes, you can machine the part to have its exact dimensions again, because the GDS bonded coating can be harder than the titanium or vanadium the part is made from."
|
|
The CGDS coating can be this hard because of the compressive stresses created when the particles impact the surface. The stresses increase the metal fatigue life, he says. This is similar to what happens in shot-peening, an industrial process similar to CGDS, but using "balls" a few millimeters in diameter to impact a surface.
|
|
"CGDS is used for very high-cost manufacture and repair, but there is no comprehensive, realistic model describing the physics of the entire process," says Jen.
|
3D with splashing
|
|
In CGDS, engineers talk about two zones. The first is the flight-zone between the spray nozzle and the surface to be sprayed. This zone was modelled by Jen in a 2005 research article in the International Journal of Heat and Mass Transfer ("Numerical investigations on cold gas dynamic spray process with nano- and microsize particles").
|
|
The second zone is the deposition zone, where the sprayed articles impact the surface. The new 3D model describes this zone.
|
|
Previous two dimensional models have attempted to solve the puzzle around CGDS bonding, but these have a severe limitation. When you extend a 2D model to 3D, you end up with a 'horizontal cylinder' descending towards the surface being sprayed.
|
|
"Unfortunately, a descending cylinder cannot model realistically enough what happens to discrete ball-shaped particles 'splashing' down in the substrate surface," says Jen.
|
|
As industry knows, the speed (velocity) the particle arrives at the substrate is critical. Too slow, and it will just bounce off. Too fast, and it may pass like a bullet through a thin substrate.
|
|
The new model animates in 3D a single spherical particle 'falling down' into the substrate metal. The substrate 'splashes up', and then the particle and substrate bond. The substrate 'splashing' looks like milk splashing up when something falls into the cat's bowl. This is called jetting behavior in industry," says Jen.
|
Cold metal, temperature rise
|
|
The model uses several parameters describing the nature of the particle and the surface: density, thermal conductivity, specific heat, melting point, elastic modulus, Poisson's ratio, Johnson-Cook plasticity and Johnson-Cook damage.
|
|
It is the first to predict in 3D how the average temperature of the particle impact zone will rise and subside, depending on the size and impact velocity of the particle. The model was published in the Journal of Thermal Spray Technology ("Particle Bonding Mechanism in Cold Gas Dynamic Spray: A Three-Dimensional Approach").
|
Just fast enough to melt
|
|
"For this 3D model, we went with the hypothesis that a metal particle has to bond with the substrate at 60% of its melting temperature, to create a strong new surface without damaging the substrate," says Professor Jen.
|
|
As an example, copper (Cu) has a melting temperature of 1083 degrees Celsius, and 60% of that is 650 degrees Celcius. So the hypothesis says that a 5 micron copper particle impacting an aluminum substrate surface, will have to be fast enough so that the average impact zone temperature goes up to at least 650 Celcius, and not much more, for good bonding to occur. According to the model, that critical impact velocity range is between 700 and 800 meters per second.
|
Supersonic energy transformation
|
|
When a copper particle travels at a supersonic speed and hits an aluminum surface, its moving (kinetic) energy is converted into heat (thermal) energy, says Prof Jen. This depends on the impact speed of the particle.
|
|
"The heat makes the particle and the impact zone 'soft and sticky', similar to melted cheese. The particle changes into a 'soft blob' that fills in the 'impact crater' in the substrate surface. At the same time, friction develops between the blob and the crater surface, which is critical to the bonding process," he says.
|
|
"The friction 'grabs' the blob, and it sinks into the substrate surface. As the blob sinks down, the molten substrate around the particle 'splashes up' in typical jetting behavior. When the jetting settles down, the bond between particle and surface is completed," says Jen.
|
Model vs the real world
|
|
The model, though limited, holds up in experimental results with copper particles sprayed onto an aluminum surface.
|
|
"When the impact velocity is within the range predicted by the model for a particle size, sufficient bonding temperature is reached and a strong CGDS coating is created. As an example, we set up our CGDS equipment in the laboratory for copper particles with an average size of 5 micron, carried by nitrogen, and impact velocity in the range of 700 to 800 meters per second deposited downwards on aluminum.
|
|
"The model predicts that at about 750 meters per second impact velocity, the critical bonding temperature of 650 degrees Celsius will be attained in the particle impact zone. In line with that prediction, we obtained excellent CGDS bonded coatings," he says.
|
|
"However, as also predicted by the model, we found with our laboratory setup that when the particle impact velocity is not within the critical range, insufficient bonding temperature is reached. This can result in poor surface coating with loosened powders and scrapping surface, which don't meet manufacturing quality standards," says Jen.
|
Grand challenge remains
|
|
The single-particle single-layer 3D model will be extended into a multi-particle, multi-layer model in follow-up projects.
|
|
Says Jen: "This 3D model is the first to describe how the temperature of the impact zone influences particle deposition. However, realistically modelling the deposition zone in CGDS remains a grand challenge to solve. In real-world conditions, particles are not of uniform size or shape, and travel at different velocities and angles. So a more complete model will have to accommodate ranges, or distributions, of all of these parameters."
|

