| Posted: Feb 15, 2018 |
Self-healing damage at the atomic level
(Nanowerk News) Scientists discovered a self-healing cycle for defects in ceramics predicted by advanced atomic-level simulations. Irradiation creates defects and causes the ordered atomic structure to become disordered. Simulations showed that creation of a threshold amount of disorder caused the displaced atoms to move faster. This sped up annihilation of the defects and healed the structure.
|
|
How atoms move in complex ceramics oxides is strongly linked to the local structure. Damage to the atomic structure results when they are exposed to irradiation or heated. How defects influence the motion of atoms over time is essential for understanding how the materials’ properties change, and how to “fix” the damage. These phenomena underpin the materials’ properties and lifetimes for radiation resistance in energy generation and containment of radioactive wastes.
|
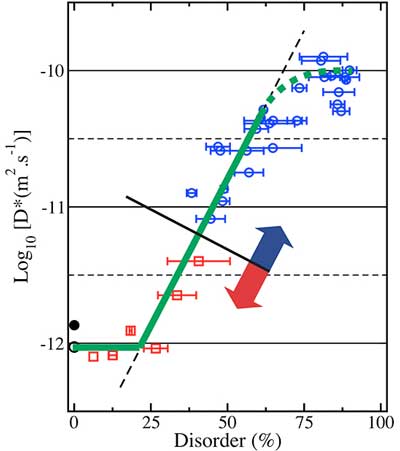 |
| Self-healing can extend the lifetime of ceramic materials subject to damage from irradiation. The irradiation processes cause the ordered atomic structure of the ceramic (red and blue circles on the left) to become disordered (right). Scientists used two computer simulation techniques to understand the subsequent atomic migration, or diffusion: standard (blue data in the graph) and accelerated (red squares and black circle) molecular dynamics. The simulations revealed a cycle of self-healing. As the disorder increased, the diffusion rate (vertical axis) remained the same until a threshold value (green arrow) was reached. After the threshold, a percolation network formed and the diffusion increased sharply. This faster diffusion led to a faster recovery of order, causing the material to self-heal. (Image: Los Alamos National Laboratory)
|
|
Diffusion in complex ceramic oxides is critical to the transport of the constituent atoms and the evolution of atomic structure due to radiation damage, sintering, and aging. In these materials, the individual atoms carry a charge that binds the structures together; negatively and positively charged ions are called anions and cations, respectively. In complex oxides which contain more than one type of cation such as pyrochlores, the migration of the ions through the atomic structure, or diffusion, and the conductivity are dramatically affected by disorder, or the way the cations are arranged in the crystal.
|
|
In particular, diffusion and conductivity are especially sensitive to cation disorder. Interestingly, this cation disorder is also at the heart of the ability of the material to maintain its crystallinity upon irradiation. This is why pyrochlores are considered as candidates to encapsulate nuclear waste. Disorder aids both conductivity and radiation resistance. However, little is known about how disorder influences cation transport.
|
|
In this study (Nature Communications, "Evidence for percolation diffusion of cations and reordering in disordered pyrochlore from accelerated molecular dynamics"), scientists investigated cation diffusion mediated by defects in the pyrochlore gadolinium titanium oxide (Gd2Ti2O7). |
|
The defects were missing atoms in the atomic structure called vacancies. Scientists used standard and accelerated molecular dynamics simulations to track atomic movements and better understand diffusion. These simulations take place over a microsecond (one-millionth of a second).
|
|
In comparison, typical atomic simulations are run to study nanoseconds (billionths of a second) of atomic movements due to the huge computational costs of running longer simulations. But with new computational techniques to simplify the dynamics of the atoms, scientists have sped up the calculations and lengthened possible times that can be investigated by these simulations.
|
|
They found that cation diffusion is slow at low levels of disorder. Once the level of disorder surpasses a threshold value, the diffusion of cations is faster. Key to this outcome was “anti-site defects.” This is where one cation (gallium, in this case) occupies a position where the other cation (titanium, in this case) is supposed to be. At a critical threshold level, the anti-site defects are essentially “touching” and create what is called a percolation network. This network allows the cations to rapidly move through the lattice. When the anti-site defects are annihilated, the structure can reorder—in essence allowing the structure to heal itself.
|
|
This healing, in turn, slows the cation diffusion. Cation diffusivity increased as the material became more disordered from irradiation and decreased as the material reordered. This cycle of self-healing is different from observations in other complex oxides and disordered models.
|
|
This research suggests a fundamentally different relationship between disorder and mass transport. These insights could improve lifetimes for complex ceramics used in applications involving extreme environments such as irradiation.
|

