| Posted: Mar 14, 2018 |
Boron can form a purely honeycomb, graphene-like 2-D structure
(Nanowerk News) Seeking for low-dimensional boron allotropes has attracted considerable interest in the past decades and tremendous theoretical works predict the existence of monolayer boron. As boron has only three valence electrons, the electron deficiency makes a honeycomb lattice of boron energetically unstable. Instead, a triangular lattice with periodic holes was predicted to be more stable.
|
|
In 2015, Prof. Wu has led a research team at Institute of Physics, Chinese Academy of Sciences, and successfully synthesized 2D borophene sheet on silver surface, which is exhibiting the predicted triangular lattice with different arrangements of hexagonal holes.
|
|
An intriguing question is whether it is possible to prepare a borophene monolayer with a pure honeycomb lattice. Honeycomb borophene will naturally host Dirac fermions and thus intriguing electronic properties resembling other group IV elemental 2D materials.
|
|
Additionally, a honeycomb 2D boron lattice may enable the superconductivity behavior. Since in the well-known high Tc superconductor, MgB2, the crystal structure consists of boron planes with intercalated Mg layers, where the boron plane has a pure honeycomb structure like graphene.
|
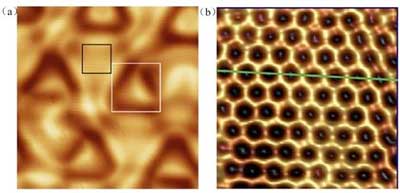 |
| High resolution STM images of borophene monolayer with honeycomb lattice on Al(111). (© Science China Press)
|
|
It is remarkable that in MgB2, superconductivity occurs in the boron planes, while the Mg atoms serves as electron donors.
|
|
Recently, the research team led by Prof. Wu reported the successful preparation of a purely honeycomb, graphene-like borophene, by using an Al(111) surface as the substrate and molecular beam epitaxy (MBE) growth in ultrahigh vacuum (Science Bulletin, "xperimental Realization of Honeycomb Borophene").
|
|
Scanning tunneling microscopy (STM) images reveal perfect monolayer borophene with planar, non-buckled honeycomb lattice similar as graphene. Theoretical calculations show that the honeycomb borophene on Al(111) is energetically stable.
|
|
Remarkably, nearly one electron charge is transferred to each boron atom from the Al(111) substrate and stabilizes the honeycomb borophene structure. This work vividly demonstrated that one can manipulate the borophene lattice by controlling the charge transfer between the substrate and the borophene. And the honeycomb borophene provides attractive possibility to construct boron-based atomic layers with unique electronic properties such as Dirac states, as well as to control superconductivity in boron-based compounds.
|

