| Posted: May 07, 2018 |
Wrap an electrode material for Li-ion battery into the inner spacing of carbon nanotube
(Nanowerk News) Researchers at the Toyohashi University of Technology have demonstrated the electrochemical performance of lithium ion batteries (LIBs) using phosphorus-encapsulated carbon nanotube electrodes, in which red phosphorus with considerable high capacity is introduced into the inner spacing of carbon nanotubes (CNTs) with a tubular structure (Journal of The Electrochemical Society, "Electrochemical Performance of Lithium Ion Battery Anode Using Phosphorus Encapsulated into Nanoporous Carbon Nanotubes").
|
|
The electrodes indicated an improvement in the electrochemical reactivity of red phosphorus when accessible pathways of lithium ions, i.e., nanopores, were formed onto the sidewalls of the CNTs where the red phosphorus was encapsulated.
|
|
Furthermore, the charge-discharge profiles and structural analysis revealed reversible electrochemical reactions and the relatively high structural stability of red phosphorus in the nanotubes even after the fiftieth charge-discharge cycle.
|
|
The charge-discharge capacities show a value two times or higher than that of graphite used in commercial LIBs. Therefore, a new electrode material for LIBs with high capacity is proposed.
|
|
Red phosphorus has attracted attention as a higher capacitive electrode material for LIBs because it can deliver a theoretical capacity approximately seven times higher than that of graphite used as a commercial electrode material for LIBs. The large difference in the capacity is thought to be due to an acceptable amount of lithium ions in the structures of graphite for LiC6 or phosphorus for Li3P.
|
|
However, red phosphorus suffers enormous volumetric changes, pulverization, and peeling off during lithium ion insertion and extraction processes, resulting in rapid capacity fading due to the decrement in the amount of electrochemically reactive red phosphorus.
|
|
Additionally, while electrons move onto the electrode during lithium ion insertion/extraction, red phosphorus has a disadvantage in terms of energy loss because of its low electronic conductivity.
|
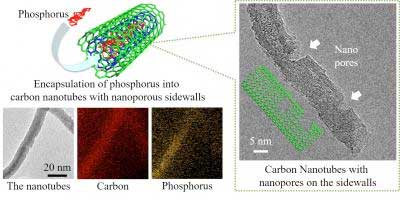 |
| Figure 1: (left) These are the elemental mapping images for phosphorus-encapsulated carbon nanotubes with nanopores on the sidewalls. (right) Transmission electron image of carbon nanotube with nanopores on the sidewalls. (Image: Toyohashi University Of Technology)
|
|
As shown in Fig. 1 (left), Tomohiro Tojo and his colleagues at the Department of Electrical and Electronic Information Engineering, Toyohashi University of Technology, have synthesized unique structures in which red phosphorus is encapsulated into the inner spacing of CNTs to prevent its peeling off from the electrode and improve its electronic conductivity.
|
|
For improving the electrochemical reactivity of red phosphorus through accessible pathways of lithium ions, nanopores (<5 nm) were also formed onto the sidewalls of the phosphorus-encapsulated CNTs as shown in Fig. 1 (right). After phosphorus encapsulation, Fig. 1 (left) shows that the phosphorus atoms were distributed inside the nanotubes, confirming the structural stability of red phosphorus.
|
|
Using phosphorus-encapsulated CNT electrodes, a reversible capacity showed approximately 850 mAh/g at the fiftieth charge-discharge cycle, as depicted in Fig. 2 (left). This was a value at least two times higher than that of graphite electrodes. Figure 2 (right) shows the estimated ratio of charge and discharge capacities (Coulombic efficiencies) of >99% after the tenth cycle and the subsequent cycles, which indicates a high reversibility of charge-discharge reactions on red phosphorus.
|
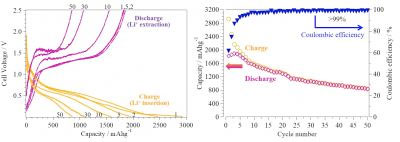 |
| Figure 2: (left) These are the charge-discharge curves for phosphorus-encapsulated carbon nanotubes with nanopores on the sidewalls. (right) Charge and discharge capacities with a ratio of charge and discharge capacities at each cycle (Coulombic efficiencies). (Image: Toyohashi University Of Technology)
|
|
However, the charge-discharge capacities gradually decreased with increasing cycle number because of the dissociation of some P-P bonds and other side reactions on the surface of phosphorus and the CNTs. Interestingly, the phosphorus-encapsulated CNT with nanopores facilitated the significant improvement in electrochemical performance compared with the phosphorus-encapsulated CNT without nanopores. This is suggested to be due to the high reactivity of red phosphorus with lithium ions through the nanopores on the sidewalls.
|
|
After the charge-discharge cycles, red phosphorus was observed to be inside the nanotubes, as is the case shown in Fig. 1 (left).
|
|
We have proposed phosphorus-encapsulated CNTs as an electrode material for LIBs with high capacity, even though additional improvements in the structures are required to achieve long-term cycling without capacity fading. Further studies will be performed on the utilization of such electrodes.
|


