| Jun 27, 2018 | |
Finding the right balance for catalysts in the hydrogen evolution reaction(Nanowerk News) Hydrogen produced from water by the hydrogen evolution reaction is an attractive clean fuel source. Production of hydrogen on a large scale at low cost is necessary to realize its viability as an alternative energy source to fossil fuels. |
|
| To achieve this goal, durable, inexpensive catalysts are required. Most present catalysts based on non-precious metals suffer from instability in the acidic solutions formed during hydrogen evolution. However, simply protecting the catalyst from the acidic solution tends to lower its activity. | |
| A collaboration led by the University of Tsukuba has recently optimized an approach to increase the stability of catalysts used in the hydrogen evolution reaction without markedly sacrificing activity. | |
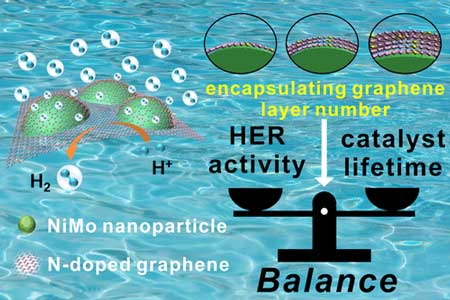
| The team found that coating catalyst nanoparticles with an optimal number of layers of graphene--a sheet of carbon atoms organized into a honeycomb lattice with high conductivity and mechanical strength--raised nanoparticle durability while allowing the nanoparticles to retain their catalytic activity. | |
| The study was reported in ACS Energy Letters ("Graphene Layer Encapsulation of Non-Noble Metal Nanoparticles as Acid-Stable Hydrogen Evolution Catalysts"). | |
 |
|
| To achieve a balance between the hydrogen evolution reaction (HER) lifetime and performance, the researchers vary the number of N-doped graphene layers (1–2, 2–3, and 3–5 layers) encapsulating NiMo nanoparticles as efficient HER catalysts and obtain the optimal number of protective layers. | |
| "We optimized the balance between the number of graphene layers coating the nanoparticles and their catalytic activity," study first author Kailong Hu says. "To do this, we had to precisely control the number of graphene layers coating the nanoparticles, which we achieved by carefully regulating the deposition time of graphene on the nanoparticles." | |
| A series of nanoparticle samples coated with different numbers of graphene layers was fabricated, characterized, and then their catalytic activity in the hydrogen evolution reaction was determined. The catalyst nanoparticles coated with the optimal number of graphene layers, which was just three to five layers, displayed similar activity in the hydrogen evolution reaction to that of an expensive platinum-based catalyst. | |
| Importantly, these nanoparticles also exhibited high stability; the graphene coating prevented the metal nanoparticles from dissolving in the acidic reaction solution. | |
| The researchers conducted theoretical calculations to support their experimental findings. The results corroborated the relationships between graphene layer number, chemical stability, and catalytic activity of the nanoparticles indicated by the experimental data. That is, the nanoparticles coated with less than three graphene layers showed higher catalytic activity than those coated with three to five layers but this came at the expense of durability; the former showed poorer chemical stability than the latter. | |
| "Our results pave the way for rational design of stable, cheap catalysts for large-scale hydrogen production at hydrogen stations by on-site polymer electrolyte membrane electrolysis under acidic conditions," co-author Yoshikazu Ito explains. | |
| The team's findings bring us a step closer to the realization of a clean sustainable future using hydrogen as a fuel source. |
| Source: University of Tsukuba | |
|
Subscribe to a free copy of one of our daily Nanowerk Newsletter Email Digests with a compilation of all of the day's news. |
