| Jul 04, 2018 | |
Porous materials shed light on environmental purification(Nanowerk News) During the last two decades, porous materials such as zeolites and metal-organic frameworks have drawn the attention of the scientific community due to the wide range of applications derived from their porosity. |
|
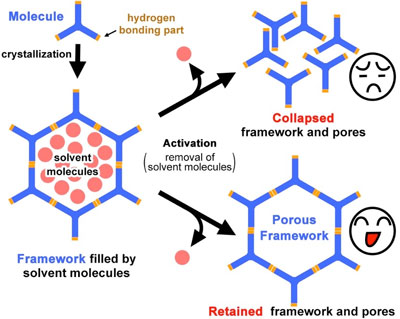
| Recently, a new class of all organic materials has emerged – the hydrogen-bonded organic frameworks (HOFs). These crystalline materials rely on two types of non-covalent interactions – π-π interactions that form the vertical stack, and hydrogen-bonding interactions that provide the order and stability between the molecular units. | |
 |
|
| Figure 1. Schematic representation of porous frameworks constructed from hydrogen molecules. The frameworks need to be stable even after the removal of solvent molecules from the pores. (Image: Osaka University) | |
| The combination of these interactions allows assembly of specifically designed molecular units to obtain ordered crystalline structures enabling the development of materials with tunable chemical and physical properties. | |
| An international collaboration between Osaka University, Japan, and the University of Castilla, Spain, developed stable single-crystalline porous hydrogen-bonded organic frameworks (HOFs) that are thermally and chemically durable and have large surface area and fluorescence properties. Through one-dimensionally stacked molecules and hydrogen-bonding, they fabricated the stable and rigid frameworks despite these frameworks consisting of weak hydrogen-bonded carboxylic acid. | |
 |
|
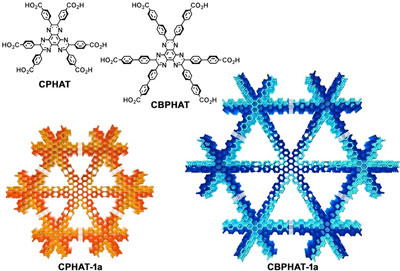
| Figure 2. Crystal structures of HOFs (hydrogen-bonded organic frameworks) composed of HAT derivatives (CPHAT and CBPHAT). (Image: Osaka University) | |
| Their research results were published in Angewandte Chemie International Edition("Docking Strategy to Construct Thermostable, Single-crystalline, Hydrogen-bonded Organic Framework with Large Surface Area"). | |
| This porous material was found to have a wide specific surface area of 1288 m2 per 1g and can keep its framework at temperatures of up to 305°C in the atmosphere. In addition, hydrogen bonds of this material are not cleaved even when immersed in alcohol or concentrated hydrochloric acid and heated. This porous material was found to have quite stable HOFs when compared to conventional materials. | |
| It is difficult to systematically form HOFs as designed, so the establishment of methodology for the formation of HOFs was sought after. This group had found that a hexaazatriphenylene (HAT) derivative with 6 carboxyphenyl groups (CPHAT) formed HOFs with high heat resistance. They thought that a HAT derivative with carboxyaryl groups was a promising molecular building block for constructing stable HOFs with a large surface area. | |
 |
|
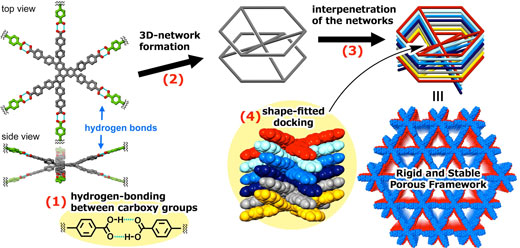
| Figure 3. Hierarchical interpretation of a rigid and stable HOF (CPHAT-1a) through (1) hydrogen-bonding between carboxy groups, (2) three-dimensional (3D) networking, (3) network interpenetration, and (4) shape-fitted docking of twisted HAT cores. (Image: Osaka University) | |
| Using a HAT derivative with carboxybiphenyl groups (CBPHAT), they obtained thermally and chemically stable HOFs with large surface area, demonstrating the effectiveness of HAT frameworks by synthesizing and crystallizing HAT derivatives that have longer hands to grasp neighboring atoms. | |
| Lead Author Ichiro Hisaki at Osaka University said, “In this study, we discovered that HAT derivatives formed rigid and stable HOFs through hydrogen-bonding between carboxy groups, three-dimensional (3D) networking, network interpenetration, and shape-fitted docking of twisted HAT cores.” |
| Source: Osaka University | |
|
Subscribe to a free copy of one of our daily Nanowerk Newsletter Email Digests with a compilation of all of the day's news. |
