| Posted: Sep 19, 2018 | |
Using donut-shaped lithium sulfide for higher performing batteries(Nanowerk News) A Korea Advanced Institute of Science and Technology (KAIST) research team developed a lithium-sulfur battery with a doughnut-shaped active material structure showing a record lifecycle of over 600 cycles (Advanced Science, "An Ultrahigh Capacity Graphite/Li2S Battery with Holey-Li2S Nanoarchitectures"). Having higher energy density and lower production cost than a lithium-ion battery (LIB), it can be used in electric vehicles that require a longer battery life. |
|
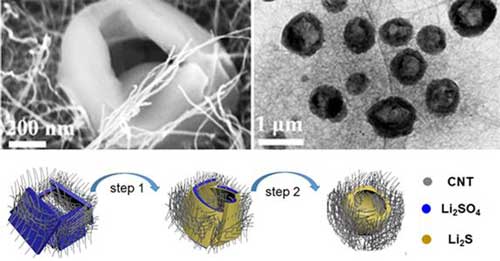 |
|
| Structural characterization of Li2SO4/CNT and Li2S/CNT electrodes and suggested mechanism for the formation of the holey-Li2S nanoarchitecture. (Image: KAIST) | |
|
There has been an intense research conducted for developing lithium |
|
| Yet, lithium sulfide is costly and, so far, there has not been an electrode architecture and electrolyte design that enables a longer lifecycle between the graphite anodes and lithium sulfide cathodes. | |
| Hence, the team produced a doughnut-shaped lithium sulfide cathode active material from low-cost lithium sulfide developed from raw materials. They have also developed a lithium sulfide ion battery with a graphite anode and lithium sulfide cathode using a high concentration salt electrolyte. | |
| This doughnut-shaped lithium sulfide showed outstanding charge and discharge reversibility through improving the transfer of lithium ions. Its highly concentrated salt electrolyte formed a stable film on the surface of the graphite electrode, which showed strong durability. | |
| Through this technology, the team achieved 30% higher energy density than that of conventional LIBs and secured a lifecycle of more than 600 cycles. This doughnut-shaped lithium sulfide-based electrode can be manufactured using low-cost raw materials and a single heat treatment process. The electrode can also be applied to existing LIBs. | |
| Professor Kim said, “We have demonstrated that applying low-cost sulfur compounds to LIBs can improve both energy density and the lifecycle simultaneously.” |
| Source: Korea Advanced Institute of Science and Technology | |
|
Subscribe to a free copy of one of our daily Nanowerk Newsletter Email Digests with a compilation of all of the day's news. |
