| Posted: Oct 09, 2018 | |
Unzip, turn around, rezip(Nanowerk News) Scientists have long known that double-stranded DNA can be unwound into single-stranded DNA by molecular motor proteins called helicases, just like pulling a zipper. But researchers at the University of Illinois have discovered that the same machinery, after changing its shape, can also put the unwound DNA back together into double-stranded DNA. |
|
| This switching between DNA rewinding and unwinding was first reported in 2015 by Illinois physics professor Y. R. Chemla. Chemla's work caught the attention of Wen Ma, a post-doctoral researcher in biological physics at Illinois, who, along with his advisor, Zaida Luthey-Schulten, advanced Chemla's work in a paper recently published in Elife ("Free-energy simulations reveal molecular mechanism for functional switch of a DNA helicase"). Ma, who completed his PhD training under the late Klaus Schulten, does computational modeling of the molecular machines that play vital roles in a cell. | |
 |
|
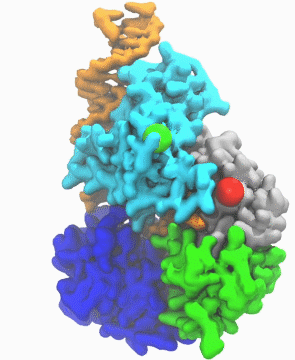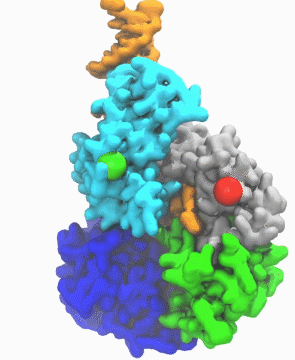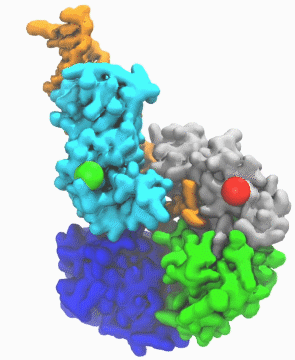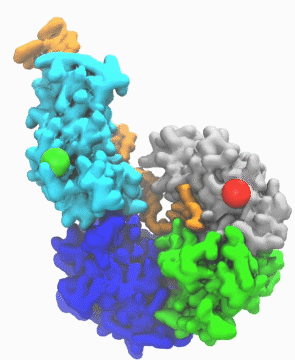
| DNA helicase switches itself from unwinding double-strand DNA to a predicted rezipping state. | |
| The unwinding is kind of conventional wisdom, says Ma. If you have something happen to the cell to cause it to mutate, such as exposure to UV light and chemicals, in response helicases will unwind the double strand into a single strand DNA, so other cell machinery may be able to come in and repair it. Obviously, he notes, sometimes you don't want every piece of double-stranded DNA to be unwound by helicases too quickly. "So there must be some control mechanism which adjusts the activity of the helicase," he says. | |
| Ma and his colleagues were intrigued by the thought that the DNA helicase—the molecular motor that drives the unwinding—could switch to rezipping, and set out to model it computationally with hopes of learning what that control mechanism was. By running the Nanoscale Molecular Dynamics (NAMD) code on the Blue Waters supercomputer at Illinois' National Center for Supercomputing Applications, Ma was able to create a simulation that included "hundreds of thousands of atoms" and study the helicase in further detail. | |
| Chemla's research team had combined two cutting-edge experimental techniques in their discovery of the unwinding-rezipping switching—simultaneous single molecule fluorescence resonance energy transfer (smFRET) and optical tweezers. smFRET is a fluorescence methodology to detect protein conformation. Optical tweezers, which apply highly focused laser light to trap microscopic objects, are used to detect helicase unwinding activity. They observed that the protein unwinds the DNA, but it doesn't go very far, until it goes back in a different conformation and rezips the DNA. But it was unclear what the molecular mechanism was that regulated this behavior. | |
| Following the footsteps of the smFRET and optical tweezers work, Ma and his colleagues used Blue Waters to model every single atom of the protein according to basic physical principles. Through this simulation, the first to provide this atomistic-level look at what is happening in the helicase, Ma and his colleagues learned the protein is really doing a kind of switching function between the two states, unwinding and rezipping, in the timescale of millisecond or longer. This long simulation time scale, according to Ma, was only able to be captured by using novel enhanced sampling simulations. | |
| "This work is important," he says, "because it enables us to link the atomic protein structure with its function. With other methodologies, like crystallography, you get a static image, and you don't know what happens dynamically. But with this simulation we learned why and how the DNA repair enzyme UvrD changes its conformation from the unzipping to the rezipping state, and we learned that the helicase tilts itself and forms stable interactions with the opposite strand once switched to rezipping. The simulation results were validated by the good agreement between the simulated smFRET efficiency for our predicted structure and the experimentally measured smFRET efficiency." | |
| Ma emphasizes that there is still much to learn about helicase function and DNA repair. The potential for applying that knowledge is huge, as the information can aid those working on targeted drug design. Perhaps one day researchers will be able to use the revealed mechanism to help restore genome stability in related cancer cells. |
| Source: By Barbara Jewett, University of Illinois at Urbana-Champaign | |
|
Subscribe to a free copy of one of our daily Nanowerk Newsletter Email Digests with a compilation of all of the day's news. |
