| Oct 01, 2019 | |
Intriguing discovery provides new insights into photoelectric effect(Nanowerk News) The discovery that free electrons can move asymmetrically provides a deeper understanding of one of the basic processes in physics: the photoelectric effect. It was first described by Albert Einstein and explains how high frequency light releases electrons from a material. The results have been published in Physical Review Letters ("Fano’s Propensity Rule in Angle-Resolved Attosecond Pump-Probe Photoionization"). |
|
| “The photoelectric effect has been studied for many years and it’s very uplifting to suddenly understand how it works in a deeper way”, says Marcus Dahlström, Associate Senior Lecturer of Mathematical Physics at Lund University in Sweden, who worked on the article with colleagues in Lund and at Stockholm University. | |
| The researchers studied how an electron that has just been released from an atom via the photoelectric effect can change its wave motion using a laser field. The free electron can both absorb and emit laser light, which changes the electron’s rotation in an asymmetrical way. | |
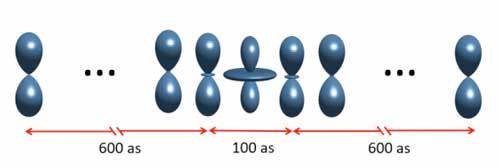 |
|
| Rapid particles in an explanatory light: When electrons are no longer fixed to the atomic shell and can move freely, their wave form can be changed using laser light. (IMage: Lund University) | |
| To discover this phenomenon, the researchers used ultrashort laser pulses with a time precision on an attosecond scale, which is staggeringly short: 0.000000000000000001 seconds. | |
| The discovery of the asymmetry in combination with the high time resolution gave the researchers the opportunity to disrupt the electrons’ ingrained behaviour. From only moving up and down along the laser field, the researchers succeeded in getting the electrons to also spread sideways. | |
| “Now that we understand there is an asymmetry in the free electrons’ movement, we can gain a better understanding of the quantum dynamics in photo-ionisation”, says David Busto, doctoral student of Atomic Physics at LTH. | |
| In classical physics, particles move in a deterministic way from one point to another via Newton’s laws. In contrast to this, quantum mechanics says that a particle can move to several places simultaneously. The researchers have been able to take advantage of the latter: | |
| “When we change the direction of the electron wave, we are using quantum mechanical interference. That is, the electron takes several paths towards its changed wave form. In classical physics the electron can only go one way.” | |
| The phenomenon of the asymmetrical movement pattern has been proved both in experiments and in theory. The results are based on the knowledge that electrons increase their rotating movements when they absorb light, something the American-Italian physicist Ugo Fano demonstrated 30 years ago. | |
| The research aims to control electrons in atoms and molecules with greater precision. In the long term, it is conceivable that this and other basic science knowledge on how atoms and molecules function will provide an opportunity to improve the way reactions are controlled in molecules, which in turn can pave the way for more effective chemistry. |
| Source: Lund University | |
|
Subscribe to a free copy of one of our daily Nanowerk Newsletter Email Digests with a compilation of all of the day's news. |
