| Mar 17, 2020 | |
Novel approach to enhance performance of graphitic carbon nitride(Nanowerk News) In a report published in NANO ("A Novel Approach to Synthesize Nitrogen-Deficient g-C3N4 for the Enhanced Photocatalytic Contaminant Degradation and Electrocatalytic Hydrogen Evolution"), scientists from the Jiangxi University of Science and Technology, Guangdong University of Petrochemical technology, Gannan Medical University and Nanchang Hangkong University in China underline the importance of defect engineering to promote catalytic performance by providing a simple and efficient way for modifying and optimizing metal-free semiconductor photocatalyst graphitic carbon nitride (g-C3N4) to solve the dual problems of environmental pollution and lack of fossil resources. |
|
 |
|
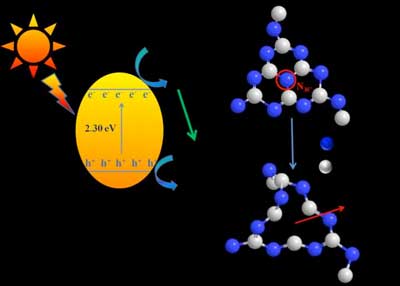
| A schematic illustration of modified g-C3N4 by nitrogen vacancies and reaction mechanism. The nitrogen vacancies not only increased the specific surface area, exposed more active sites, but also inhibited the recombination of photogenerated carriers on the surface of photocatalysts, resulting in the enhancement of photocatalytic and electrocatalytic performances. (Image: Kai Yang) | |
| With the rapid growth of industrialization and population, environmental pollution and shortage of fossil resources have become two major challenges for sustainable social development in the 21st century. Hence, the development of green treatment technology is an imperative. | |
| Semiconductor photocatalysis technology has become one of the most promising strategies due to its green, nontoxic and high efficiency by using solar energy. Recently, graphitic carbon nitride (g-C3N4), as a new semiconductor photocatalyst of nonmetallic polymer, has attracted wide attention in photo-catalytic field due to its better stability and optical properties. | |
| The photocatalytic activity of bare g-C3N4 is unsatisfactory due to its smaller surface area and rapid recombination of photogenerated carriers under visible light irradiation. | |
| In this work, urea was used as the activated support for the nitrogen vacancies on the basis of bare g-C3N4 by the calcination of melamine. This led to great improvement of photocatalytic performance for the degradation of organic dyes in water, such as rhodamine (RhB), acid orange II, methyl orange (MO) and methyl blue (MB) under visible light irradiation (λ > 420nm). Thus, electrocatalytic performance for hydrogen evolution was achieved due to broader light response, efficient generation and migration of electron/hole charge carriers. | |
| It is hoped that this research will provide an idea about the innovative design, synthesis and fabrication of modifying g-C3N4 and other N-based photocatalysts. There is potential for applying this catalyst to the treatment of environmental pollutants and the preparation of new energy. |
| Source: World Scientific | |
|
Subscribe to a free copy of one of our daily Nanowerk Newsletter Email Digests with a compilation of all of the day's news. |
