| Jul 31, 2020 | |
Way, shape and form: Synthesis conditions define the nanostructure of manganese dioxide(Nanowerk News) Scientists at Tokyo Institute of Technology explore a novel and simplistic method to synthesize manganese dioxide with a specific crystalline structure called β-MnO2. Their study sheds light on how different synthesis conditions can produce manganese dioxide with distinct porous structures, hinting at a strategy for the development of highly tuned MnO2 nanomaterials that could serve as catalysts in the fabrication of bioplastics. |
|
 |
|
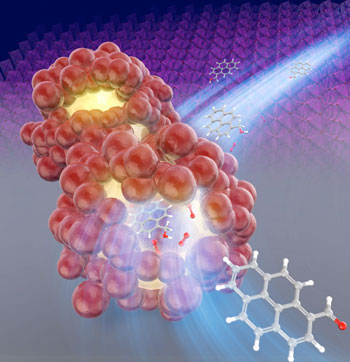
| Acceleration of the chemical reaction by β-MnO2 catalyst in the nanospace of the particles. (Image: Keiko Kamata, Tokyo Institute of Technology) | |
| Materials engineering has advanced to a point at which not only are we concerned about the chemical composition of a material, but also about its structure at a nanometric level. Nanostructured materials have recently drawn the attention of researchers from a variety of fields and for good reason; their physical, optical, and electrical characteristics can be tuned and pushed to the limit once methods to tailor their nanostructure are available. | |
| Manganese dioxide (chemical formula MnO2) nanostructured metal oxide that can form many different crystalline structures, with applications across various engineering fields. | |
| One important use of MnO2 is as a catalyst for chemical reactions, and a particular crystalline structure of MnO2, called β-MnO2, is exceptional for the oxidation of 5-hydroxymethylfurfural into 2,5-furandicarboxylic acid (FDCA). | |
| Because FDCA can be used to produce environment-friendly bioplastics, finding ways to tune the nanostructure of β-MnO2 to maximize its catalytic performance is crucial. | |
| However, producing β-MnO2 is difficult compared with other MnO2 crystalline structures. Existing methods are complicated and involve the use of template materials onto which β-MnO2 "grows" and ends up with the desired structure after several steps. | |
| Now, researchers from Tokyo Institute of Technology led by Prof. Keigo Kamata explore a template-free approach for the synthesis of different types of porous β-MnO2 nanoparticles. | |
| Their method, described in their study published in ACS Applied Materials & Interfaces ("Template-Free Synthesis of Mesoporous β-MnO2 Nanoparticles: Structure, Formation Mechanism, and Catalytic Properties"), is outstandingly simple and convenient. | |
| First, Mn precursors are obtained by mixing aqueous solutions and letting the solids precipitate. After filtration and drying, the collected solids are subjected to a temperature of 400 °C in a normal air atmosphere, a process known as calcination. During this step, the material crystallizes and the black powder obtained afterwards is more than 97% porous β-MnO2. | |
| Most notably, the researchers found this porous β-MnO2 to be much more efficient as a catalyst for synthesizing FDCA than the β-MnO2 produced using a more widespread approach called the "hydrothermal method." | |
| To understand why, they analyzed the chemical, microscopic, and spectral characteristics of β-MnO2 nanoparticles produced under different synthesis conditions. | |
| They found that β-MnO2 can take on markedly different morphologies according to certain parameters. In particular, by adjusting the acidity (pH) of the solution in which the precursors are mixed, β-MnO2 nanoparticles with large spherical pores can be obtained. This porous structure has a higher surface area, thus providing better catalytic performance. | |
| Excited about the results, Kamata remarks: "Our porous β-MnO2 nanoparticles could efficiently catalyze the oxidation of HMF into FDCA in sharp contrast with β-MnO2 nanoparticles obtained via the hydrothermal method. Further fine control of the crystallinity and/or porous structure of β-MnO2 could lead to the development of even more efficient oxidative reactions." | |
| What's more, this study provided much insight into how porous and tunnel structures are formed in MnO2, which could be key to extending its applications, as Kamata states: "Our approach, which involves the transformation of Mn precursors into MnO2 not in the liquid-phase (hydrothermal method) but under an air atmosphere, is a promising strategy for the synthesis of various MnO2 nanoparticles with tunnel structures. These could be applicable as versatile functional materials for catalysts, chemical sensors, lithium-ion batteries, and supercapacitors." | |
| Further studies like this one will hopefully allow us to one day harness the full potential that nanostructured materials have to offer. |
| Source: Tokyo Institute of Technology | |
|
Subscribe to a free copy of one of our daily Nanowerk Newsletter Email Digests with a compilation of all of the day's news. |
