| Mar 02, 2021 |
On-surface synthesis of graphene molecules and their superlattices
(Nanowerk News) National University of Singapore scientists have devised a new method for the synthesis of nanographene molecules with a high product yield for the development of next generation quantum devices (Science Advances, "Ultrahigh-yield on-surface synthesis and assembly of circumcoronene into a chiral electronic Kagome-honeycomb lattice").
|
|
On-surface chemical reactions have shown promising potential in the synthesis of new organic functional materials such as atomically-precise nanographenes. The core concept of this strategy relies on the rational design of specific molecular precursors, which subsequently undergo chemical transformation along certain reaction pathway towards the desired product.
|
|
The electronic, magnetic and optical properties of these nanographene molecules can be precisely tuned for the development of next-generation quantum devices. Unfortunately, conventional surface-assisted synthetic routes often involve a series of cascade reactions with competing reaction pathways. This inevitably leads to the formation of numerous undesired products and lowers the yield.
|
|
The limited yield of the targeted products poses a challenge for practical applications of the nanographenes.
|
|
A NUS research team led by Prof Jiong LU, in collaboration with Prof Jishan WU’s research group, both from the Department of Chemistry, NUS has developed a route for synthesizing the hexagonal zigzag-edged nanographene, known as circumcoronene, on a copper (111) substrate.
|
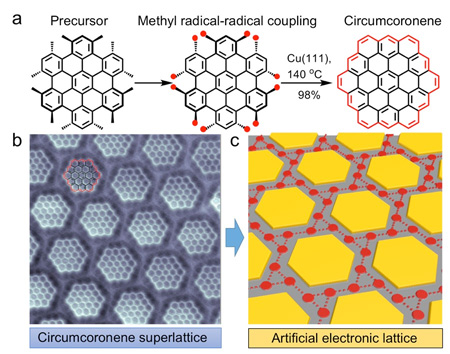 |
| Figure shows the transformation from the precursor molecule to the atomically-precise circumcoronene superlattice. (a) The precursor molecule undergoes a cyclodehydrogenation chemical process with the use of copper (111) substrate which forms the circumcoronene. (b) High-resolution image of the circumcoronene superlattice obtained by using a non-contact atomic force microscopy with a cobalt tip. (c) Schematic illustration of the chiral Kagome-honeycomb lattice. (© Science Advances)
|
|
The reaction route relies on the robust dehydrogenative coupling of the methyl groups at the adjacent sites of the rationally-designed precursor molecules, followed by the ring closure reactions on the metallic substrate. This forms the elusive circumcoronene molecule consisting of 19 fused benzene rings (Figure (a)).
|
|
Importantly, such a synthetic route allows for an ultra-high yield of the reaction product (up to 98%), which has not been attained to date.
|
|
The electrostatic interactions between the large number of circumcoronene molecules and the copper substrate enabled the molecules to self-assemble into extended superlattices. This was observed by the team using bond-resolved scanning probe microscopy measurements (Figure (b)).
|
|
The researchers demonstrate that the unique hexagonal zigzag topology of circumcoronenes, along with their periodic electrostatic landscape, confines the two-dimensional (2D) electron gas on the copper (111) surface. This creates a chiral electronic Kagome-honeycomb lattice with two emergent electronic flat bands (Figure (c)). |
|
This arrangement of the circumcoronene molecules in a regular grid of hexagons and triangles can be particularly interesting in a wide range of condensed matter physics because of their favourable potential in the realisation of a variety of exotic many-body phenomenon, including anomalous quantum Hall states, Wigner crystallisation, and topological insulating transitions.
|
|
Prof Lu said, “Our findings open up a new route for the ultra-high yield synthesis of nanographene and atomically-precise fabrication of synthetic two-dimensional lattices with unique electronic properties for future technological applications.”
|

