| Jan 18, 2023 |
RNA lipid nanoparticle engineering stops liver fibrosis in its tracks, reverses damage
(Nanowerk News) Since the success of the COVID-19 vaccine, RNA therapies have been the object of increasing interest in the biotech world. These therapies work with your body to target the genetic root of diseases and infections, a promising alternative treatment method to that of traditional pharmaceutical drugs.
|
|
Lipid nanoparticles (LNPs) have been successfully used in drug delivery for decades. FDA-approved therapies use them as vehicles for delivering messenger RNA (mRNA), which prompts the cell to make new proteins, and small interfering RNA (siRNA), which instruct the cell to silence or inhibit the expression of certain proteins.
|
|
The biggest challenge in developing a successful RNA therapy is its targeted delivery. Research is now confronting the current limitations of LNPs, which have left many diseases without an effective RNA therapy.
|
|
Liver fibrosis occurs when the liver is repeatedly damaged and the healing process results in the accumulation of scar tissue, impeding healthy liver function. It is a chronic disease characterized by the buildup of excessive collagen-rich extracellular matrix (ECM). Liver fibrosis has remained challenging to treat using RNA therapies due to a lack of delivery systems for targeting activated liver-resident fibroblasts. Both the solid fibroblast structure and the lack of specificity or affinity to target these fibroblasts has impeded current LNPs from entering activated liver-resident fibroblasts, and thus they are unable to deliver RNA therapeutics.
|
|
To tackle this issue and help provide a treatment for the millions of people who suffer from this chronic disease, Michael Mitchell, J. Peter and Geri Skirkanich Assistant Professor of Innovation in the Department of Bioengineering, and postdoctoral fellows Xuexiang Han and Ningqiang Gong, found a new way to synthesize ligand-tethered LNPs, increasing their selectivity and allowing them to target liver fibroblasts.
|
|
Lulu Xue, Margaret Billingsley, Rakan El-Mayta, Sarah J. Shepherd, Mohamad-Gabriel Alameh and Drew Weissman, Roberts Family Professor in Vaccine Research and Director of the Penn Institute for RNA Innovation at the Perelman School of Medicine, also contributed to this work.
|
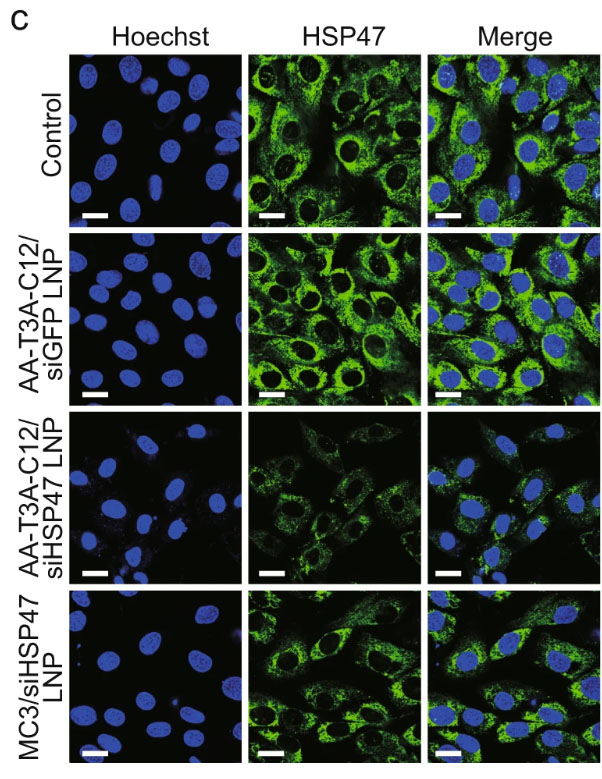 |
| Immunofluorescence staining shows different ligand-tethered LNPs’ abilities to knock down HSP47 in activated fibroblasts. (Image: University of Pennsylvania)
|
|
Their study, published in Nature Communications ("Ligand-tethered lipid nanoparticles for targeted RNA delivery to treat liver fibrosis"), shows how a small-molecule ligand incorporated into the synthesis of the ionizable lipid, a key component of the LNP, creates an affinity to the notoriously hard-to-target activated fibroblasts in the liver responsible for the buildup of collagen.
|
|
The collagen buildup is accompanied by an increased expression of Heat Shock Protein 47 (HSP47), the protein that drives collagen biogenesis and secretion. Overexpression of HSP47 and increased collagen biogenesis ultimately progresses to fibrosis.
|
|
Once their LNPs arrive at and enter the target cell, siRNA is released, which silences the expression of HSP47, inhibits the production of collagen and stops fibrosis in its tracks. The treatment, shown to be successful in mice, is a promising treatment for liver fibrosis in humans.
|
|
This novel approach to ionizable lipid synthesis is the key to opening many more doors for RNA therapy to treat diverse diseases.
|
|
“To make LNPs selective enough to target hepatic stellate cells, those that drive fibrosis, we incorporated an anisamide ligand, a molecule which has a high affinity for the receptor on these stellate cells, into the structure of the ionizable lipid,” says Mitchell. “Essentially, we created a lock-and-key mechanism to target and unlock delivery to these hard-to-reach cells.”
|
|
The synthesis process was developed by Han and colleagues as a “one-pot, two-step” process. To create a library of ionizable lipids, the team first put an anisamide ligand (AA) precursor and different amino cores together. They then added the hydrophobic tail to create AA-tethered ionizable lipids. Anisamide was chosen as the ligand due to its neutral and stable nature as well as its affinity for the overexpressed sigma receptors on stellate cells. Once the library of AA-tethered LNPs was created, the team analyzed their abilities to target and deliver therapy to cells through a two-round selection process.
|
|
“We needed to find a specific AA-tethered LNP that was both potent and selective,” says Han. “The first round of the selection process was done by examining how well our LNPs could knock down green fluorescence protein (GFP) in fibroblasts to measure potency. GFP provides great visual evidence for how therapeutic RNA turns off gene expression in real time.”
|
|
“In the second round, we tested the selective ability of the potent LNP,” says Han. “We did this by blocking the sigma receptor to understand how significant the specific AA ligand group was in the LNPs ability to get into target cells. Unsurprisingly, we showed that the AA group was significant; after the sigma receptor blockade, we lost the lock-and-key mechanism and the AA-tethered LNP would not enter the target cell.”
|
|
The team identified AA-T3A-C12 as both a potent and selective LNP carrying therapeutic siRNA able to achieve 65% knockdown of HSP47 expression in mice as well as enhance the recovery of damaged liver tissue. The results of the study conclude that the AA-T3A-C12 LNP outperforms the MC3 LNP, a clinically utilized non-viral vector that has been FDA-approved for use in hepatic, or liver, cell RNA therapy.
|
|
This new ligand-tethered LNP provides a form of treatment for liver fibrosis and the synthesis method provides a way to tailor LNPs to other previously hard-to-target cells and tissues in the body.
|
|
“The potential of LNPs is enormous,” says Han. “We’re making LNPs smarter and more efficient.”
|
|
“We are excited to have produced a potential treatment that tackles the genetic root of this liver disease,” says Mitchell. “And, because this LNP delivery vehicle works in fibrotic cells of the liver, it may lead to developing a treatment for other types of fibrosis in the body, such as fibrosis that arises in the lung or in tumors.”
|
|
“Beyond what we have investigated in the liver, this method of creating LNPs can be used to unlock delivery for therapies to other cell types,” he adds. “We could potentially target cells in the brain, lungs or heart by installing specific targeting ligands into the ionizable lipid structure. There are many avenues from here and we are excited to continue pushing this research in new directions.”
|

