| Jun 19, 2023 |
Organic Light-emitting diodes: Making the blue shine brighter and longer
(Nanowerk News) Organic light-emitting diodes are used in smart phones, tablets, and large TV screens. As they do not need any additional background illumination, they are energy-efficient. The can be produced at low costs by thin-film technology and also work on flexible carrier materials, thus enabling bendable displays and variable room illumination solutions.
|
|
An OLED consists of two electrodes, of which at least one is transparent. Thin layers of organic semiconducting materials are located between them. Their light results from electroluminescence. When applying an electrical field, electrons from the cathode and holes (positive charges) from the anode are injected into the organic materials that act as emitters. There, electrons and holes meet and form electron-hole pairs. When these pairs collapse into their initial state again, they release energy that is used by the organic materials to emit light. All colors can be obtained by mixing blue, green, and red.
|
 |
| Emission within and between the molecules makes deep-blue OLEDs shine more efficiently and stably. (Image: Zhen Zhang)
|
Why Blue Is Difficult
|
|
So far, phosphorescent OLEDs in red and green have been available for commercial applications only. Blue light can be produced by fluorescent OLEDs for a short time only. Blue OLEDs have difficulties to combine a high efficiency with a high luminescence and a long service life. Blue pixels are fainter or faint more quickly than green and red pixels.
|
|
In cooperation with researchers from Shanghai University, researchers from KIT’s Institute of Organic Chemistry (IOC) and Institute of Biology and Chemical Systems – Functional Molecular Systems (IBCS-FMS) have now developed a new strategy for the production of highly efficient and stable deep-blue organic light-emitting diodes (Science Advances, "Efficient deep-blue luminescence based on dual-channel intra/intermolecular exciplexes").
|
|
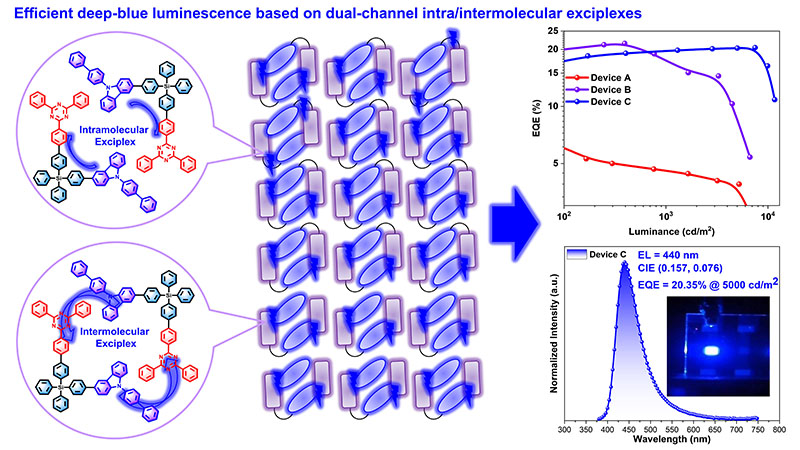
The researchers produced a new type of molecule, in which carbazole and triazine fragments are linked by a silicon atom (CzSiTrz). When the molecules assemble to form nanoparticles, electronic excitation causes intramolecular charge transfer emission and intermolecular exciplex luminescence, resulting in a dual-channel intra-/intermolecular exciplex emission.
|
|
An exciplex is an electronically excited molecule complex. Its emission differs from the emissions of excited single molecules. “The exciplex strategy allows to achieve deep-blue electroluminescence, because the energy levels of the electron-donating carbazole fragments and electron-accepting triazine fragments can be adjusted independently of each other,” says Professor Stefan Bräse from IOC and IBCS-FMS.
|
New OLEDs Reach a High External Quantum Efficiency and High Luminance
|
|
The team succeeded in producing deep-blue OLEDs with a record-setting external quantum efficiency of 20.35 percent. The external quantum efficiency is the ratio between the radiation output and power input. Moreover, these OLEDs reach a high luminance of 5000 candela per square meter (cd/m2). The perceivable blue has the coordinates of 0.157/0.076 on the chromaticity diagram of the International Commission on Illumination (CIE).
|
|
“Easy synthesis of the molecule and production of the components pave the way for a new generation of efficient and long-lived deep-blue OLEDs,” Bräse points out.
|

