| Nov 24, 2023 |
Programmable DNA origamis enable targeted drug delivery
(Nanowerk News) For decades, researchers have sought improved control over the distribution and activity of therapeutic drugs to minimize adverse side effects while maximizing efficacy. Most approved drugs like small molecules and monoclonal antibodies operate systemically with little innate precision over where they accumulate in the body and how long they persist. This lack of precision triggers toxic reactions and discourages high dosing, constraining progress.
|
|
Recently, breakthroughs in structural DNA nanotechnology have opened an entirely new path to exert spatial governance over drug behavior using self-assembling programmable nanostructures with defined shapes and chemistry. Unlike previous passive drug carriers like liposomes and polymersomes, these intelligent DNA systems feature modular programmability, precise nanoscale geometry, and readily tunable surface moieties that confer directionality.
|
|
In a new study published in Scientific Reports ("Biodistribution and function of coupled polymer‑DNA origami nanostructures"), researchers demonstrate the power of coupling disease-targeting aptamers to polyethylene glycol (PEG)-stabilized DNA nanostructures to concentrate and sustain therapeutic activity within target tissue sites. The researchers explored multiple rod-shaped nanostructures with varying size and aspect ratios, discovering that longer particles above 100 nm persisted significantly longer in vivo while diffusing away from injection sites more slowly.
|
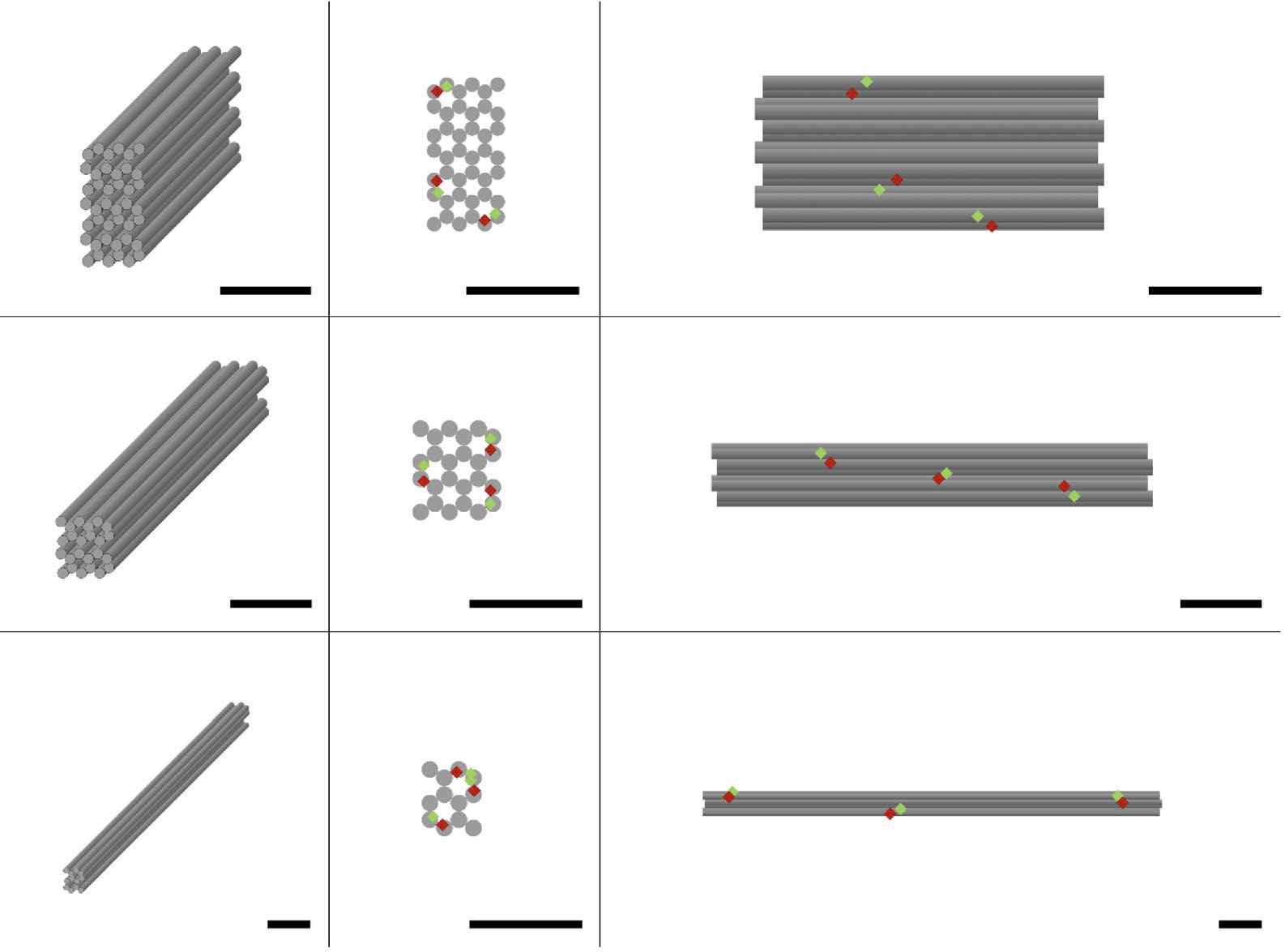 |
| Structural characterization of the DNA origami nanostructures and quality assessment of their assembly. (a) Design schematic. The rows show the different DNA origami nanostructures investigated: cuboid, short rod and long rod (from top to bottom). The columns show different views of the DNA origami nanostructures: 3D, front and side view (from left to right).FRET pairs are distributed evenly on the DNA origami nanostructures and shown as red and green diamonds. All scale bars are 20 nm. (© NPG)
|
|
Attaching aptamers directed against the inflammatory cytokine TNF-alpha conferred anti-inflammatory effects in mouse models of delayed type hypersensitivity reactions (DTHR). Mice treated with TNF-alpha-neutralizing DNA nanostructures showed reduced tissue swelling, lower immune cell infiltration, and less epidermal thickening comparable to anti-TNF-alpha monoclonal antibodies. Yet unlike conventional antibodies dispersed systemically throughout the body, the aptamer nanostructures remained concentrated in target tissue.
|
|
This localization effect focuses therapeutic impact at disease sites while avoiding systemic exposure, thereby increasing the therapeutic window. It also reduces the dose needed for equivalent treatment outcomes. The modularity of these programmable nanostructures further enables personalized optimization and multiplexing by facile substitution of targeting and therapeutic moieties.
|
|
The findings highlight the disruptive potential of self-assembling DNA nanostructures as intelligent drug carriers with inherent spatial control. As costs continue falling exponentially, such precision DNA systems could replace mainstream technologies like monoclonal antibodies for improved specificity while accelerating development of synergistic aptamer cocktails, tissue-localized drug delivery, and personalized medicine tailored to individual genetics and disease phenotypes.
|

