| Posted: January 17, 2007 |
Nanotechnology batteries - the end of exploding batteries? |
|
(Nanowerk News) A new, safer type of Li-Ion nanobattery that might help prevent future fires and explosions related to conventional Li-Ion battery use has been developed by researchers at Tel Aviv University. These nanobatteries should also prove useful for various micro devices used for medical, military and a range of other applications.
|
|
A brief history of batteries and the way they work
|
|
Although ancient batteries go back as early as 250 BC, this knowledge bares very little relation on the development of batteries in modern times. The term battery was coined by Benjamin Franklin in the mid 18th Century to describe a device called the Leyden jar, which was an array of charged storage devices, or capacitors. Later that century, Luigi Galvani developed a device that could produce an electric current by chemical means far greater than the current produced by earlier electrostatic generators. This was a circuit consisting of two dissimilar metals in contact, their other ends exposed to salt water. The device developed by Galvani often called a galvanic cell or a voltaic cell was further developed by Alessandro Volta a few years later. Volta piled up a series array of galvanic cells to invent what was later called a voltaic pile. Today many still refer to a battery as a pile.
|
|
A battery converts chemical energy directly into electrical energy. It consists of one or more voltaic cells, each of which is composed of two half cells. Each cell has a positive terminal and a negative terminal; these do not touch each other but are immersed in a solid or liquid electrolyte that contains ions that can react with chemicals in the electrode. Chemical energy is converted into electrical energy by chemical reactions that transfer charge between the electrode and the electrolyte at their interface. Such reactions are called faradaic and are responsible for current flow through the cell. Ordinary, non-charge-transferring (non-faradaic) reactions also occur at the electrode-electrolyte interfaces. Non-faradaic reactions are one reason that voltaic cells (particularly the lead-acid cell of ordinary car batteries) "run down" when sitting unused.
|
|
Lithium ion (or Li-Ion) batteries are one of the most common types of batteries used in today's consumer electronic products ranging from mobile phones to laptop computers. The origins of lithium batteries date back to the beginning of the 20th Century, but turning the technology into a useful commercial product capable of hundreds of recharge cycles took long, frustrating decades which ended with Sony's introduction of the first commercial version in the early 1990s. One of the major problems with Li-Ion batteries is safety. To allow usage of Li-Ion batteries by the general public, several mandatory safety mechanisms had to be built into the battery. These include a shut-down contraption against overheating, a tear-away tab for internal pressure, a special vent built for pressure relief, and a thermal interrupt against overcharging. These devices make the battery larger, more expensive, and are constantly at risk of malfunction.
|
|
Recently several large computer companies including Dell, Apple, Toshiba, Lenovo, and Sony ordered a recall of millions of Li-ion batteries used on laptop computers after a series of fires and explosions that happened following uncontrolled overheating of the batteries. Sony, the battery manufacturer, admitted that the explosions were caused by a short circuit inside a battery cell caused by a metallic contamination of the lithium-ion chemicals. Although the number of batteries that caught on fire or exploded was small, the financial damage to the companies and the manufacturer is measured in the hundred of millions of dollars.
|
 |
| Dell laptop explodes in Japan (Image: RescueHumor.com)
|
|
Three-dimensional Nanobatteries – The end of exploding batteries?
|
|
About four years ago, Professors Emanuel Peled and >Diana Golodnitsky from the School of Chemistry at Tel Aviv University and Professor Menachem Nathan from the Department of Electrical Engineering-Physical Electronics also at Tel Aviv University began working on a new type of battery technology. This new technology allows for the production of smaller, powerful batteries, but even more importantly, batteries that have very little risk of overheating or exploding.
|
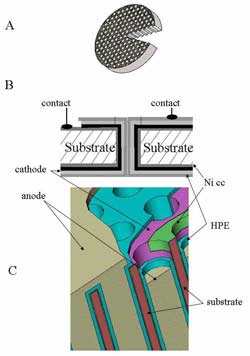 |
| Schematic design of the nanobattery
|
|
The batteries developed by the team from Tel Aviv University (TAU) work in a different way than conventional batteries. Using a silicon or glass substrate, the team from TAU created a matrix of tiny holes each 50 microns in diameter and 500 micron deep. Each of these holes functions as an independent micro battery or microchannel with an output power of around 10 microW. The power of a 1 cm2 3D nanobattery is about 150-200mW.
|
|
One of the most important aspects of this new technology compared to existing battery types is its safety. Since each nanobattery is comprised of thousands of small batteries, even if one of these small batteries has a short circuit and fails, the entire battery can keep functioning, lossing only a very small amount of power. Similar damage to a conventional Li-Ion battery could result in substantial loss of power or a complete malfunction and in extreme cases even fire or explosion.
|
|
The new nanobattery technology has several potential applications:
|
|
RFID batteries - Radio Frequency Identification (RFID) technology was first demonstrated by researchers from Los Alamos, California in 1973. The most familiar form of RFID is the RFID tag. There are passive, semi-passive, and active RFID chips. Passive RFID chips use the minute electrical current induced in their antennas by incoming radio frequency signals to provide them with just enough power to operate and transmit data in response. Semi-active and active RFID tags include a battery, which helps them to stay on, respond more quickly, and transmit their information more effectively; the trade-off is that when the battery drains, it needs to be replaced or the tag will be unable to function properly. While passive RFID tags have a typical range of a few meters, active RFID tags can reach dozens or even hundreds of meters. These ranges make them perfect for monitoring product tracking, transport payments, wireless sensors, and various other applications. Nanobatteries can be very useful as small high power active RFID batteries.
|
|
MEMS batteries - Micro-Electro-Mechanical Systems or MEMS are electronic and mechanical devices several microns in size used in the medical and pharmaceutical industry (for example in implantable medial devices), automotive industry, as parts for projectors, optical switching, printers and various sensors.
|
|
Existing thin-film "microbatteries" that may be useful in MEMS systems include planar (2D) thin-film batteries, such as those developed at Oak Ridge National Laboratories by a group led by John Bates. Each planar thin-film battery is composed of successive sub-micron to several micron-thick layers of cathode (LiCoO2 or LiMn2O4), lithium phosphorus oxynitride (LIPON) electrolyte and a lithium or silicon tin oxynitride anode. Planar batteries have cathodes with thicknesses of up to 5 micron, a capacity of 0.133 mAh/cm2 and a "geometrical" energy density of 0.28 mAh/cm2. These batteries require a large footprint, and therefore are limited in their usefulness for MEMS. The solution developed by the team from Tel Aviv University was to move to 3D thin-film configurations in which the reduction of the footprint area is compensated by an increase in area in the third (height) dimension. In this way the new nanobatteries were able to reach 20-30 times the capacity achieved by previous micro battery designs enabling a whole new range of future MEMS applications. Some 3D microbattery concepts, proposed by other research groups, involve preparation of complicated architectures based on carbon posts or a graphite mesh. This is a challenging task with poor reproducible structures.
|
|
Batteries for smart dust – Smartdust is concept for a wireless MEMS sensors that can detect anything from light and temperature to vibrations. Using a large amount of sensors is not a new concept - the U.S. military experimented with the idea as early as the Vietnam War (Operation Igloo White). While the older sensors were relatively large and only somewhat effective, Professor Christopher Pister from UC Berkeley suggested in 2001 to create a new type of micro sensor that could theoretically be as small as a grain of sand. Research into this idea is ongoing and DARPA (the Defense Advanced Research Projects Agency) is funding the work. If these sensors will be developed successfully, they will require tiny, powerful batteries to operate and transmit their data. The nanobatteries developed at Tel Aviv University might be able to supply the necessary power while keeping the size of the sensor as small as possible.
|
|
Batteries for consumer applications - In the future, the new nanobattery technology could be implemented in consumer applications and be used as replacements for commercial Li-Ion batteries for smaller, lighter, and safer laptops, mobile phones, and cameras.
|
|
This has been an abbreviated version of an article that appeared on the The Future of Things website.
|


