| Posted: January 19, 2007 |
Nanoparticles bond like atoms in a molecule |
|
(Nanowerk News) Nanoparticles are typically well-rounded individuals - to the extent that it is difficult to involve them in any interactions that depend on geometrical preferences. Researchers at the Massachusetts Institute of Technology (MIT), US, have now succeeded in breaking the symmetry of nanoparticles using a mathematical principle known as the hairy ball theorem ("Divalent Metal Nanoparticles").
|
|
This theorem states that on a sphere covered in fur one cannot brush all the hairs flat without creating at least two whirls, technically known as singularities. Accordingly, Francesco Stellacci's team turned gold nanoparticles into tiny furballs by depositing self-assembling thiol monolayers onto their surfaces. Using transmission electron microscopy, the researchers could show that the thiols arrange themselves in parallel rings, like the latitude lines on a globe. The singularities manifest themselves as single molecules of thiol sticking out from each of the poles. Unlike the molecules in the rings, the polar ones are poorly stabilized by their neighbours and thus easier to displace.
|
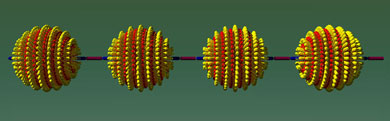 |
| Nanoparticles link up. (Image: Science)
|
|
The researchers managed to replace the polar thiols with longer chain 'handles' carrying carboxylic acid groups at the other end. They polymerized these constructs by applying the widely known reaction that is used in the production of nylon.
|
|
They dissolved the coated nanoparticles in toluene and brought them into contact with an aqueous solution of 1,6 diaminohexane. The polymer of nanoparticles could be harvested from the phase boundary, just like nylon. Group leader Stellacci explained: 'We have created the nanoscale equivalent of polymers. We expect that these materials will show the validity of some of the fundamental assumptions of polymer physics and present a plethora of new properties.'
|
|
Nanotech pioneer Cees Dekker from the Technical University of Delft, Netherlands, welcomed the new development. 'This is very original work that may open a new avenue for using nanoparticles,' he told Chemistry World. 'It adds a new functionality to them, namely the ability to bond to other particles in specific directions, just like atoms [that] make up molecules.'
|

