| Posted: December 23, 2009 |
Thanks to nanoparticles, DNA detection by the naked eye is now possible |
|
(Nanowerk News) A sensitive yet uncomplicated method to detect differences in DNA strands using metal nanoparticle solutions has been developed by Roejarek Kanjanawarut and Xiaodi Su at the Institute of Materials Research and Engineering at A*STAR, Singapore ("Colorimetric Detection of DNA Using Unmodified Metallic Nanoparticles and Peptide Nucleic Acid Probes"). The method requires no modification of the surfaces of the nanoparticles, making it particularly fast and versatile to implement.
|
|
The researchers worked on the principle that aggregated and dispersed nanoparticles have different optical properties that make the solutions take on different colors. This means that the results of their tests can be displayed in minutes, and recorded qualitatively by the naked eye and quantitatively by a standard spectrometer.
|
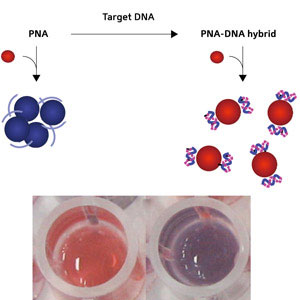 |
| Schematic diagram showing the conversion of PNA to a PNA–DNA complex (top) and photographs (bottom) of gold nanoparticle solutions with the addition of a PNA–DNA complex (left) and PNA (right).
|
|
The tendency of nanoparticles to aggregate in solution has been considered a drawback, and in previous approaches to use them for sensing, DNA strands were directly attached to the particles’ surfaces to prevent them clumping together. Kanjanawarut and Su turned the tables to take advantage of the natural propensity to aggregate as inspiration for their assays. Compared with the earlier nanoparticle- and chip-based DNA assays, the new method is cost-effective as it involves no time-consuming surface modifications, particle bioconjugation, biohazardous labeling or tedious assay procedures, explains Su.
|
|
The researchers used gold and silver nanoparticles that have a coating of negatively charged molecules. These charges repel one another, preventing the nanoparticles from aggregating in solution. They then added peptide nucleic acid (PNA) probes to the solution, which bound to the negative surfaces, shielding the charge and allowing the nanoparticles to agglomerate. The solution with gold nanoparticles changed from bright red to dark purple.
|
|
The PNA can be tailored to bind selectively to whichever DNA strand is the target for detection. After adding the target DNA strand to the solution, it bound to the PNA, making a PNA–DNA complex. The solution remained bright red, clearly different from the aggregated dark purple solution (Fig. 1). Even one mismatched DNA base compared to the target strand could be detected.
|
|
The color difference results from the formation of the PNA–DNA complex on the nanoparticle surface, which is negatively charged due to the negative charge born by the DNA chains; this makes the nanoparticles repel one another and thus take longer to aggregate.
|
|
Su hopes that their approach can be used to measure specific DNA sequences for diagnosis or fundamental research. “The sequence involved in this study, for example, is from a human gene. Single-base-mismatch detection in this gene is associated with, and thus markers for, autoimmune diseases such as psoriasis,” she says.
|

