| Posted: May 10, 2010 |
Neutron scattering unravels the structure of aromatic 'pi-pi' interactions |
|
(Nanowerk News) Aromatic interactions arise from delocalised π-electrons, and they play a key role in a very wide range of natural and industrial processes. For example protein folding, DNA and RNA base stacking, protein-nucleic acid recognition, drug design and oil recovery. However, detailed knowledge of the structures adopted by simple model aromatic molecules is currently lacking, and is essential to our fundamental understanding of π-π interactions.
|
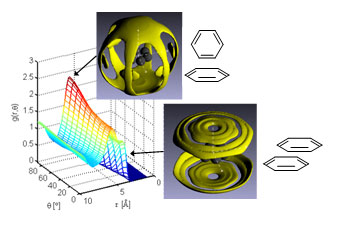 |
| Figure 1: The aromatic structure of liquid benzene, with spatial and orientational correlations showing the dominant “Y-shaped” and “parallel-displaced” motifs.
|
|
Researchers from the London Centre for Nanotechnology and the ISIS Neutron Scattering Facility (STFC Rutherford Appleton Laboratory) have now obtained definitive structural data for two archetypical aromatic liquids: benzene and toluene. By judiciously labelling these molecules with different isotopes of hydrogen, they were able to use neutron scattering to unravel the spatial and orientational packing of these molecules ("Structure ofπ-π Interactions in Aromatic Liquids").
|
|
The results identify a configuration that is now termed “Y-shaped” as a dominant motif in aromatic liquids. The near absence of the “T-shaped” geometry, previously proposed as the most favourable, is likely to lead to a significant re-evaluation of aromatic interactions in liquids.
|
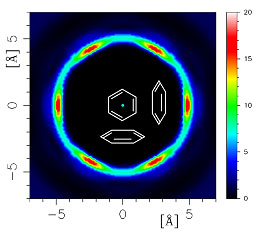 |
| Figure 2: a cut through the six-fold lantern showing that “Y-shaped” interactions are strongly preferred to “T-shaped”.
|
|
The experiments were conducted on the SANDALS instrument at ISIS, and the work was sponsored in part by the Natural Environment Research Council UK (NERC).
|


