| Posted: May 10, 2010 |
Untangling the quantum entanglement behind photosynthesis |
|
(Nanowerk News) The future of clean green solar power may well hinge on scientists being able to unravel the mysteries of photosynthesis, the process by which green plants convert sunlight into electrochemical energy. To this end, researchers with the U.S. Department of Energy (DOE)’s Lawrence Berkeley National Laboratory (Berkeley Lab) and the University of California (UC), Berkeley have recorded the first observation and characterization of a critical physical phenomenon behind photosynthesis known as quantum entanglement.
|
|
Previous experiments led by Graham Fleming, a physical chemist holding joint appointments with Berkeley Lab and UC Berkeley, pointed to quantum mechanical effects as the key to the ability of green plants, through photosynthesis, to almost instantaneously transfer solar energy from molecules in light harvesting complexes to molecules in electrochemical reaction centers. Now a new collaborative team that includes Fleming have identified entanglement as a natural feature of these quantum effects. When two quantum-sized particles, for example a pair of electrons, are “entangled,” any change to one will be instantly reflected in the other, no matter how far apart they might be. Though physically separated, the two particles act as a single entity.
|
 |
| Mohan Sarovar (seated) and (from left) Akihito Ishizaki, Birgitta Whaley and Graham Fleming carried out the first observation and characterization of quantum entanglement in a real biological system.
|
|
“This is the first study to show that entanglement, perhaps the most distinctive property of quantum mechanical systems, is present across an entire light harvesting complex,” says Mohan Sarovar, a post-doctoral researcher under UC Berkeley chemistry professor Birgitta Whaley at the Berkeley Center for Quantum Information and Computation. “While there have been prior investigations of entanglement in toy systems that were motivated by biology, this is the first instance in which entanglement has been examined and quantified in a real biological system.”
|
|
The results of this study hold implications not only for the development of artificial photosynthesis systems as a renewable non-polluting source of electrical energy, but also for the future development of quantum-based technologies in areas such as computing – a quantum computer could perform certain operations thousands of times faster than any conventional computer.
|
|
“The lessons we’re learning about the quantum aspects of light harvesting in natural systems can be applied to the design of artificial photosynthetic systems that are even better,” Sarovar says. “The organic structures in light harvesting complexes and their synthetic mimics could also serve as useful components of quantum computers or other quantum-enhanced devices, such as wires for the transfer of information.”
|
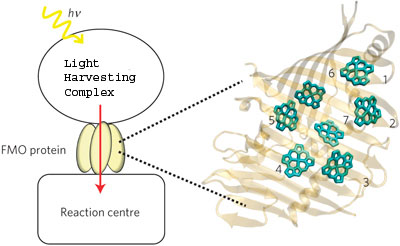 |
| This schematic shows the absorption of light by a light harvesting complex and the transport of the resulting excitation energy to the reaction center through the FMO protein. On the right is a monomer of the FMO protein, showing its orientation relative to the antenna and the reaction center. The numbers label FMO’s seven pigment molecules. (Image from Mohan Sarovar)
|
|
What may prove to be this study’s most significant revelation is that contrary to the popular scientific notion that entanglement is a fragile and exotic property, difficult to engineer and maintain, the Berkeley researchers have demonstrated that entanglement can exist and persist in the chaotic chemical complexity of a biological system.
|
|
“We present strong evidence for quantum entanglement in noisy non-equilibrium systems at high temperatures by determining the timescales and temperatures for which entanglement is observable in a protein structure that is central to photosynthesis in certain bacteria,” Sarovar says.
|
|
Sarovar is a co-author with Fleming and Whaley of a paper describing this research that appears on-line in the journal Nature Physics titled “Quantum entanglement in photosynthetic light-harvesting complexes.” Also co-authoring this paper was Akihito Ishizaki in Fleming’s research group.
|
|
Green plants and certain bacteria are able to transfer the energy harvested from sunlight through a network of light harvesting pigment-protein complexes and into reaction centers with nearly 100-percent efficiency. Speed is the key – the transfer of the solar energy takes place so fast that little energy is wasted as heat. In 2007, Fleming and his research group reported the first direct evidence that this essentially instantaneous energy transfer was made possible by a remarkably long-lived, wavelike electronic quantum coherence.
|
|
Using electronic spectroscopy measurements made on a femtosecond (millionths of a billionth of a second) time-scale, Fleming and his group discovered the existence of “quantum beating” signals, coherent electronic oscillations in both donor and acceptor molecules. These oscillations are generated by the excitation energy from captured solar photons, like the waves formed when stones are tossed into a pond. The wavelike quality of the oscillations enables them to simultaneously sample all the potential energy transfer pathways in the photosynthetic system and choose the most efficient. Subsequent studies by Fleming and his group identified a closely packed pigment-protein complex in the light harvesting portion of the photosynthetic system as the source of coherent oscillations.
|
|
“Our results suggested that correlated protein environments surrounding pigment molecules (such as chlorophyll) preserve quantum coherence in photosynthetic complexes, allowing the excitation energy to move coherently in space, which in turn enables highly efficient energy harvesting and trapping in photosynthesis,” Fleming says.
|
|
In this new study, a reliable model of light harvesting dynamics developed by Ishizaki and Fleming was combined with the quantum information research of Whaley and Sarovar to show that quantum entanglement emerges as the quantum coherence in photosynthesis systems evolves. The focus of their study was the Fenna-Matthews-Olson (FMO) photosynthetic light-harvesting protein, a molecular complex found in green sulfur bacteria that is considered a model system for studying photosynthetic energy transfer because it consists of only seven pigment molecules whose chemistry has been well characterized.
|
|
“We found numerical evidence for the existence of entanglement in the FMO complex that persisted over picosecond timescales, essentially until the excitation energy was trapped by the reaction center,” Sarovar says.
|
|
“This is remarkable in a biological or disordered system at physiological temperatures, and illustrates that non-equilibrium multipartite entanglement can exist for relatively long times, even in highly decoherent environments.”
|
|
The research team also found that entanglement persisted across distances of about 30 angstroms (one angstrom is the diameter of a hydrogen atom), but this length-scale was viewed as a product of the relatively small size of the FMO complex, rather than a limitation of the effect itself.
|
|
“We expect that long-lived, non-equilibrium entanglement will also be present in larger light harvesting antenna complexes, such as LH1 and LH2, and that in such larger light harvesting complexes it may also be possible to create and support multiple excitations in order to access a richer variety of entangled states,” says Sarovar.
|
|
The research team was surprised to see that significant entanglement persisted between molecules in the light harvesting complex that were not strongly coupled (connected) through their electronic and vibrational states. They were also surprised to see how little impact temperature had on the degree of entanglement.
|
|
“In the field of quantum information, temperature is usually considered very deleterious to quantum properties such as entanglement,” Sarovar says. “But in systems such as light harvesting complexes, we see that entanglement can be relatively immune to the effects of increased temperature.”
|


