| Posted: June 18, 2010 |
Classical thought experiment on molecular motors brought to life in granular gas |
|
(Nanowerk News) Researchers from the University of Twente, the University of Patras in Greece and the Foundation for Fundamental Research on Matter (FOM) have for the first time experimentally realised, almost a century later, an idea dating from 1912. In that year the physicist Smoluchowski devised a prototype for an engine at the molecular scale in which he thought he could ingeniously convert Brownian motion into work.
|
|
The team of scientists have now successfully constructed this device at the much larger scale of a granular gas. Moreover, they have shown that an intriguing exchange takes place between the vanes of the engine and the granular gas: once the vanes have started rotating, they in turn induce a rotating motion in the gas, a so-called convection roll. This reinforces the movement of the device and allows for a virtually continuous rotation. FOM PhD student Peter Eshuis and his colleagues published their results on 16 June 2010 online in the renowned journal Physical Review Letters ("Experimental Realization of a Rotational Ratchet in a Granular Gas").
|
|
Molecular motors, such as those responsible for tensing and relaxing your muscles, move in a strange manner: they propel themselves forwards despite - or thanks to - a continuous bombardment of the randomly moving molecules in their surroundings. This random movement is called Brownian motion and a well-constructed motor at the nanoscale actually makes use of this to generate a directed movement (and therefore work).
|
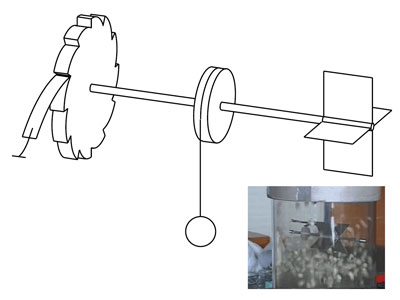 |
| Figure 1. Smoluchowski's thought experiment. Smoluchowski's thought experiment with the vanes on the right, the cog on the left and in the middle a pulley with a weight. Inset: our granular demonstration experiment.
|
|
The device introduced by the physicist Marian Smoluchowski in 1912, as a thought experiment, is a classical example of such a motor (see Figure 1). It consists of a series of vanes mounted on an axis, which are set in motion under the influence of the molecular bombardment. As this motion would take place in both rotational directions, Smoluchowski devised a second element, an asymmetrical cog. This would ensure that the axis could only rotate in a single direction and could therefore perform work, for example by pulling a small weight up. However, in 1963 Richard Feynman demonstrated that the second law of thermodynamics would prevent the device from working in a system that was in a state of thermal equilibrium and with this, the thought experiment appeared to have been consigned to the waste bin.
|
|
Yet the objection formulated by Feynman does not apply to a system far removed from a thermal equilibrium, such as a granular gas. Researchers from the University of Twente, the University of Patras and FOM have now successfully demonstrated that Smoluchowski's thought experiment works superbly in this environment (see experiment in Figure 2).
|
 |
| Figure 2. The thought experiment is brought to life. The thought experiment is brought to life in a granular gas: the experimental setup (left) and the device in operation (right).
|
|
Brownian motion
|
|
Imagine that you are driving your car through a storm with hailstones as big as footballs. Every time that such a hailstone hits you, the impact propels you forwards, backwards or sideways, with the result that you stagger forwards across the road like a drunkard. A far-fetched example? Not on the molecular scale: there (due to the continual collisions with the surrounding molecules) all particles move in this manner, a phenomenon termed Brownian motion.
|
|
Granular gas
|
|
If a container filled with beads is vigorously vibrated on top of a shaking device, the beads move so fast that a gaseous state of rapidly moving beads arises. In many ways this state is similar to the molecular gaseous state. However, there exists one major difference with a molecular gas: when you stop shaking, the beads will lose their energy in a very short space of time and come to lie motionless on the bottom of the container. This happens because a bit of energy is lost in each collision between two beads. A constant supply of energy is therefore needed to maintain the granular gaseous state and this explains why this system remains far from thermal equilibrium.
|


