| Posted: August 2, 2010 |
For the first time, atomic force microscopy helps scientists reveal the exact chemical structure of a natural compound |
|
(Nanowerk News) In a pioneering research project, for the first time, scientists at IBM and the University of Aberdeen have collaborated to "see" the structure of a marine compound from the deepest place on the Earth using an atomic force microscope (AFM). The results of the project open up new possibilities in biological research which could lead to the faster development of new medicines in the future.
|
|
Molecule from the deep seaLast year, scientists from the University's Marine Biodiscovery Centre began work on a species of bacterium from a mud sample taken from the Mariana Trench — the deepest place on Earth located 10,916 meters (35,814 feet) beneath the Pacific Ocean. This pressure-tolerant bacterium — called Dermacoccus abyssi — produced a chemical compound which could not be recognized.
|
|
Using a technique called noncontact atomic force microscopy (AFM), scientists from IBM Research were able to image individual molecules with atomic resolution within one week. These images together with density functional theory calculations confirmed the identification as cephalandole A, which is actually known and originally isolated from a Taiwanese orchid.
|
|
"Sourcing unique chemical compounds from some of the Earth's most extreme regions and identifying the structure of these compounds can be an extremely time-consuming process," said Leo Gross, scientist, IBM Research - Zurich. "This technique demonstrates that scanning probe microscopes can add powerful functionality and speed in identifying the structure of molecules which are challenging to resolve with conventional techniques."
|
|
The experiment was the first successful use of an AFM in the determination of, what was at the time, an unknown molecular structure.
|
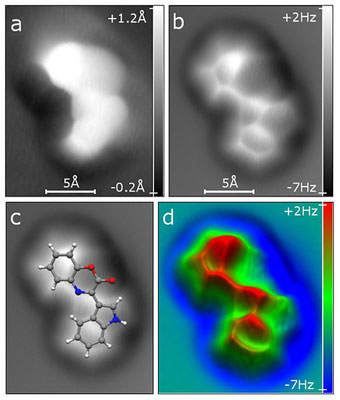 |
| SPM measurements of the unknown compound. STM and AFM measurements with a CO-functionalized tip were performed on a specimen adsorbed on the NaCl(2 ML)/Cu(111) surface. Image size: 16 Å × 19 Å. a, Constant-current STM measurement (tunnelling current I ¼ 1.2 pA, sample voltage V ¼ 150 mV). b–d, Constant-height AFM measurement (oscillation amplitude A ¼ 0.5 Å, sample voltage V ¼ 0 mV). The images show the original raw data (b), the same image with the molecular model of cephalandole A (1) overlaid as a guide to the eye (c) and a low-pass filtered three-dimensional representation (d).
|
|
"The Earth's natural environment is rich with a diverse range of unique organisms from which a vast array of chemical compounds can be sourced, many of which are entirely unknown to science," said Professor Marcel Jaspars, Director of the Marine Biodiscovery Centre at the University of Aberdeen. "These compounds have the potential to be used in the development of pharmaceuticals and other novel biomedical products. But in order to harness this potential we must first understand these compounds in terms of their molecular structure in order to determine whether they are viable for use in medicine."
|
|
For hundreds of years scientists have understood that a wide-range of unique resources — in the form of chemical compounds — exist in the natural environment, such as in oceans and deserts, which have the potential to be used in the development of new medicines.
|
|
Motivated by this insight, scientists at the Marine Biodiscovery Centre at the University of Aberdeen are focusing, in particular, on harnessing the potential of marine organisms as a source for the discovery of chemical compounds, which could be used to develop new treatments for cancer, inflammation, infection, and parasitic diseases.
|
|
Using high-resolution mass spectrometry, scientists at the University of Aberdeen quickly identified the chemical composition of the compound, but determining its exact molecular structure was more challenging. Even the use of state-of-the-art nuclear magnetic resonance techniques would not allow them to determine the exact structure due to the small number of protons and the positioning of certain atoms in the compound.
|
|
The scientists were left with four potential structures, none of which could be ruled out by the nuclear magnetic resonance data alone. The only remaining possibility to find the correct structure would be to take a chemical synthesis of the proposed structures, which is a very complex task that can take several months.
|
|
"Determining the structure of an unknown compound is a time-consuming process which could take months, therefore the ability to immediately 'see' the structure of a chemical compound simply by looking through a microscope is a tremendous feat," said Professor Jaspars. "This new approach could lead to much faster identification of unknown compounds and ultimately speed up the process of the development of new medicines."
|
|
The findings were reported in the online August 1 issue of the peer-reviewed, scientific journal Nature Chemistry.
|

