| Posted: August 16, 2010 |
Breakthrough nanoparticle gene therapy prevents retinal degeneration |
|
(Nanowerk News) In one of only two studies of its kind, a study from researchers at Tufts University School of Medicine and the Sackler School of Graduate Biomedical Sciences at Tufts demonstrates that non-viral gene therapy can delay the onset of some forms of eye disease and preserve vision. The team developed nanoparticles to deliver therapeutic genes to the retina and found that treated mice temporarily retained more eyesight than controls. The study, published online in advance of print in Molecular Therapy ("POD Nanoparticles Expressing GDNF Provide Structural and Functional Rescue of Light-induced Retinal Degeneration in an Adult Mouse"), brings researchers closer to a non-viral gene therapy treatment for degenerative eye disorders.
|
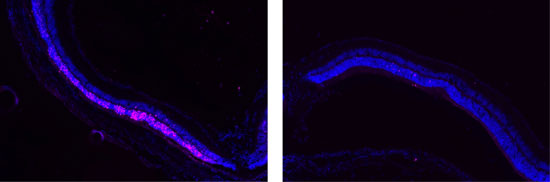 |
| Image on left shows damage (pink) to the retina. Image on right show that POD GDNF nanoparticles protected the retina from damage.
|
|
"Our work shows that it is possible to attain therapeutic results using non-viral gene delivery methods, specifically, nanoparticles. Nanoparticles, which are small enough to penetrate cells and stable enough to protect DNA, are capable of preventing retinal cell death and preserving vision," said senior author Rajendra Kumar-Singh, PhD, associate professor of ophthalmology at Tufts University School of Medicine (TUSM) and member of the genetics; neuroscience; and cell, molecular, and developmental biology program faculties at the Sackler School of Graduate Biomedical Sciences at Tufts.
|
|
"The most common approach to gene therapy involves using a virus to deliver DNA to cells. While viruses are very efficient carriers, they can prompt immune responses that may lead to inflammation, cancer, or even death. Non-viral methods offer a safer alternative, but until now, efficiency has been a significant barrier," said Kumar-Singh.
|
|
In a model simulating the progression of human retinal degeneration, the researchers treated mice with nanoparticles carrying a gene for GDNF (Glial Cell Line-Derived Neurotrophic Factor), a protein known to protect the photoreceptor cells in the eye. Retinas treated with the GDNF-carrying nanoparticles showed significantly less photoreceptor cell death than controls. Preservation of these cells resulted in significantly better eyesight in the treatment group seven days after treatment, compared to controls.
|
|
The protection conferred by the GDNF-carrying nanoparticles was temporary, as tests fourteen days after treatment showed no difference in eyesight between treated mice and controls.
|
|
"The next step in this research is to prolong this protection by adding elements to the DNA that permit its retention in the cell. Bringing forth a more potent and enduring result will move us closer to clinical application of non-viral gene therapy," said Kumar-Singh.
|
|
AMD, which results in a loss of sharp, central vision, is the number one cause of visual impairment among Americans age 60 and older. Retinitis pigmentosa, an inherited condition characterized by night blindness and loss of peripheral vision, affects approximately 1 in 4,000 individuals in the United States.
|
|
Additional authors on the study are first author Sarah Parker Read, an MD/PhD candidate at TUSM and Sackler and member of Kumar-Singh's lab, and Siobhan Cashman, PhD, research assistant professor in the department of ophthalmology at TUSM and member of Kumar-Singh's lab.
|
|
In a previous study, this same team of researchers developed the gene delivery method used in this research. The researchers showed that a peptide called PEG-POD, which compacts DNA into nanoparticles, delivers genes to the retina more efficiently than other non-viral carriers.
|
|
This study was supported by grants from The Ellison Foundation; the National Eye Institute, part of the National Institutes of Health; the Virginia B. Smith Trust; and grants to the Department of Ophthalmology at Tufts University from the Lions Eye Foundation and Research to Prevent Blindness. Sarah Parker Read is part of the Sackler/TUSM Medical Scientist Training Program, which is funded by the National Institute of General Medical Sciences, part of the National Institutes of Health.
|

