| Jan 07, 2011 |
Anything goes in oxides
|
|
(Nanowerk News) Researchers in Japan have demonstrated why the material Sr2IrO4,—a transition metal oxide—that was expected to be an electrical conductor is actually an insulator ("Microscopic Study of a Spin-Orbit-Induced Mott Insulator in Ir Oxides"). Harnessing this material's unusual conducting properties could form the basis for novel electronic devices or superconductors.
|
|
The difference between an electrical conductor and an insulator is that electrons in the latter cannot move freely through the crystal. This is because insulators have a gap in their energy spectrum that electrons cannot overcome. Hiroshi Watanabe, Tomonori Shirakawa and Seiji Yunoki from the RIKEN Advanced Science Institute in Wako and the Japan Science and Technology Agency have now uncovered how the electronic gap in Sr2IrO4 arises. Other RIKEN scientists had shown previously that the compound is an insulator.
|
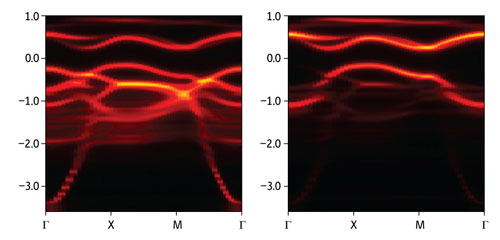 |
| Figure 1: The energetic states of Sr2IrO4. The left panel shows the spectrum of all the computed electronic states. A gap in energy (y-axis) is clearly visible for electrons moving in all crystallographic directions (x-axis). The letters on the x-axis denote specific crystallographic directions. The right panel shows an isolation of the gap arising from states that form as a result of the spin-orbit interaction.
|
|
Sr2IrO4 is a member of the oxygen-containing compounds based on transition metals that have high atomic numbers. In these transition metals, the electrons of elements such as nickel, copper or cobalt strongly interact with each other, which results in effects such as superconductivity or magnetism.
|
|
In compounds made from the heavier transition metals, the outermost electrons circle the atoms in the so-called '5d electron shell', which is relatively distant from the core. For electrons that occupy this shell there is an unusually strong interaction between their magnetic property, called spin, and the orbital motion around the atomic nucleus. The energy of this spin–orbit interaction is as large as the electron's energy of motion or the energy arising from the electrostatic interaction between the electrons. This has dramatic consequences on their electronic properties, according to Yunoki, who led the research team. "Literally anything can happen in 5d electron systems because of the subtle balance of those three fundamental energy scales."
|
|
How this energetic interplay modifies the electron conducting behavior in Sr2IrO4 became evident from the researchers' calculations. The strong spin–orbit interaction in Sr2IrO4 shifts some of the electronic states to higher energies, which is sufficiently strong to create an energy gap in the electronic states (Fig. 1).
|
|
Furthermore, the calculations reveal an intriguing connection to the family of high-temperature superconductors that have a similar gap in their electronic states. In these compounds, superconductivity is achieved through a small addition of atoms introducing an electron surplus. The researchers are now investigating the possibility that this could also be the case here. "It would have an enormous impact if one can make Sr2IrO4 superconducting," says Yunoki. "We hope that our theoretical calculations will be of help to experimentalists."
|

