| Jan 19, 2011 |
Researchers synthesize giant molecule
|
|
(Nanowerk News) Producing molecules comparable with large bio-molecules in size, shape and structure is an age-old dream of organic chemists. An international research team led by A. Dieter Schlüter from the Department of Materials Science at ETH Zurich has now succeeded in synthesising the biggest macromolecule to date ("The Largest Synthetic Structure with Molecular Precision: Towards a Molecular Object").
|
|
Building almost endlessly long chain molecules by chemically linking together small molecules is easy. However, constructing rods, spheres, fibrils, membranes and other defined 3-dimensional structures with a stable shape – something that is a matter of routine in nature and is achieved in every simple virus – has, until now, presented unsolvable problems for science. This is because length alone is still not enough to yield a three-dimensional shape.
|
|
A challenge for polymer chemist Dieter Schlüter: "To produce a thick polymer you can't just festoon monomers with huge branching units and join them together." The 3-dimensional (so-called steric) hindrance would be too large.
|
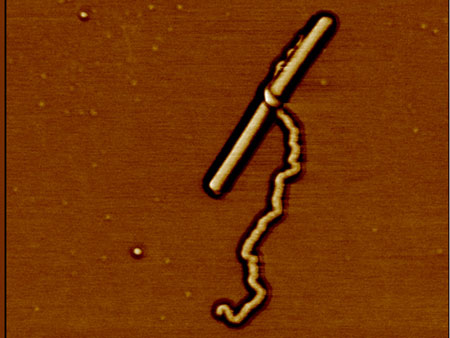 |
| Atomic force microscope image of the giant molecule, in this case accidentally "hugging" a tobacco mosaic virus, which it nearly resembles in size and shape.
|
|
The solution is a procedure inspired by a natural process: dendritic (tree-like) growth. Monomers carrying only very small branchings are linked together linearly to form the base polymer (PG1) – the trunk, so to speak. The starting material for this is a methyl methacrylate. The resulting polymer backbone consists on average of 10,600 repeat units, each carrying two terminal amines, making a total of 21,200. These amines are like offshoots, onto each of which identical branching units (dendrons) are grafted for further branching. Four "growth phases" produce spaghetti-shaped objects with a molecular mass of 200 million Dalton and a diameter of 10 nanometres.
|
|
Synthesis with molecular precision
|
|
The shape and thickness of the dendronised polymers are a consequence of their step-by-step construction, since the innumerable twigs branch more with each reaction step. Accordingly, however, the number of chemical linking reactions running in parallel must also double with each step. Thus in the fourth growth phase there are already 170,000 amidations taking place – simultaneously on a single molecule.
|
|
To ensure that no defects occur, the linking reactions must take place as completely as possible, i.e. on almost every one of the polymer's 10,600 repeat units. Otherwise weak points are formed, which cannot be tolerated. Schlüter compares it to "A Viennese sausage with a notch in the middle. The polymer would bend easily at this point, as a result of which its mechanical properties would change."
|
|
But the build-up was carried out with high precision. By choosing appropriate reaction conditions, post-doctoral student Baozhong Zhang achieved a conversion of 94.3 percent over all consecutive steps (from the PG1 base polymer to PG5).
|
|
Applications in engineering and medicine
|
|
Practically speaking, PG5 could be used, for example, as a valve for molecular machines. "If the terminal amines of the thick polymer chain are charged positively or negatively, the chain contracts as the salt concentration of the surrounding aqueous medium increases. And a decrease causes it to elongate again," explains Schlüter. The nano-objects would be usable as drug release systems in the field of medicine, since active ingredients can easily be attached by covalent or supramolecular forces to each of the 170,000 terminal amino groups present in a closely packed arrangement, like leaf and flower buds on the fine polymer twiglets.
|
|
However, PG5 is also a rich playground for other modifications, because almost any kind of chemistry can be carried out at the "buds". Schlüter is currently interested in one aspect in particular: "All the textbooks say that the properties of a polymer depend on chain length and chain length distribution. 'Thick' chains have not existed until now. We want to find out whether thickness has any effect on the stiffness and elasticity of the material, for example." This is currently being investigated in collaboration with colleagues in Geneva. The next thing Schlüter's team want to do is to test how far it is possible to push the game of branching dendronised polymers. "Will we succeed with a PG6? A PG7? Where's the limit?"
|

