| Posted: June 19, 2007 |
Stretching a molecule helps it conduct |
|
(Nanowerk News) Chemists in Taiwan have shown that the conductance of a molecule fluctuates as it is stretched.
|
|
Many researchers believe that a molecule's conformation, or structure, influences its electron transporting properties. Chun-hsien Chen and Tien-Yau Luh of National Taiwan University and colleagues have now measured this effect experimentally ("The effect of molecular conformation on single molecule conductance: measurements of π-conjugated oligoaryls by STM break junction").
|
|
The researchers designed pi-conjugated molecular wires of varying size, made of alternating benzene and furan groups. Pi-conjugated molecules have alternating single and multiple bonds and delocalised electrons.
|
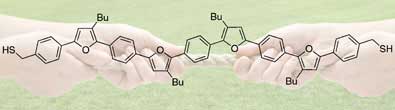 |
| Stretching out a molecular wire helps electrons move along its length
|
|
Chen and Luh's team measured the conductance of their wires in a gold-molecule-gold assembly using scanning tunnelling microscopy (STM). To make the gold-molecule-gold bridge, a gold STM tip was moved toward a gold substrate in a solution containing the molecules. Once the connection was made, the tip was moved away, stretching the molecule until the junction between it and the gold broke. Chen found that in some measurements, the conductance increased just before the gold-molecule-gold bridge snapped. They attribute this partly to the molecule stretching and improving its conjugation conformation, which in turn improves the movement of the electrons and thus conductance.
|
|
'Compared to previous studies, the molecules here are flexible and highly conjugated,' said Chen, 'which is the key to making this phenomenon apparent.'
|
|
They also showed that the resistance at the molecule-gold contact was smaller with their larger molecules, and say that the way in which the groups at the ends of the molecule interact with the orbitals of the other groups in the molecule is also important.
|
|
'This is an important observation,' said Richard Nichols, who also studies the conductance of molecular wires using STM at the University of Liverpool, UK. 'Learning to precisely control and stabilise the electrical properties of small groups of molecules or even individual molecules is an important future goal in molecular electronics.'
|

