| May 26, 2011 |
Improving DNA sequencing: Sponge-like biosensor crams enormous power into tiny space
|
|
(Nanowerk News) Vanderbilt University engineers have created a "spongy" silicon biosensor that shows promise not only for medical diagnostics, but also for the detection of dangerous toxins and other tiny molecules in the environment. This innovation was originally designed to detect the presence of particular DNA sequences, which can be extremely helpful in identifying whether or not a person is predisposed to heart disease or certain kinds of cancer. The new sensor is described in the Optical Society's open access journal, Optics Express ("Guided mode biosensor based on grating coupled porous silicon waveguide").
|
|
Biological chemical sensors save lives by detecting dangerous substances in the environment or specific molecules in the blood that could signal life-threatening diseases. Current sensor technologies, however, are limited because of their large size compared to the extremely minute sizes of some of the chemicals to be detected. In most cases, when attempting to sense something very small with a large sensor, the small molecules don't perturb the sensor's properties enough for detection.
|
|
As Vanderbilt University's Xing Wei, a graduate student, and Sharon M. Weiss, an associate professor of electrical engineering and physics, report in Optics Express, it's possible to eliminate this challenge by making sensors with features that are comparative in size to the molecules being detected, greatly increasing the sensitivity of current sensing technology.
|
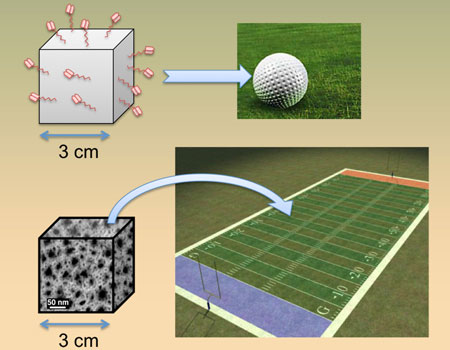 |
| This image illustrates the enormous increase in surface area possessed by porous silicon sensors. If we assume a traditional sensor can capture molecules on an area equivalent to that of a golf ball, then a similarly sized porous silicon sensor has enough surface area to capture molecules on an area equivalent to that of a football field. The ability to capture more molecules increases the probability and sensitivity of detecting small molecules such as DNA or toxins.
|
|
To do this, the engineers turned to a porous silicon material, which acts essentially as a small sponge that can then be "seeded" or filled with all sorts of substances that change its properties—resulting in a detector that's highly sensitive to small molecules. Capturing a particular sequence of DNA involves seeding the sensor with a single strand of DNA, so that only the complementary strand can attach to it and everything else gets rinsed away.
|
|
Why use a porous silicon material? Weiss and Wei stress the significance of their new sensor's enormous surface area relative to its small size. To illustrate this point, Weiss describes two cubes, one of which is 3 cm on a side, with a flat surface that DNA can be attached to—providing 54 cm2 of available surface area to attach these molecules. The other is an identical-size cube, made into a porous silicon sponge and with the ability to access the volume of the cube with all of the internal surface area, providing a surface area that is nearly 10,000 times greater than the first cube. This is like comparing the area of a golf ball to the area of a football field—with the same footprint (See image). "It's an enormous increase in the potential of how many molecules you can capture, and this is one reason why our new sensor is so much more sensitive for detecting small molecules," says Weiss.
|
|
And by using a "grating," a type of surface texturing of the porous silicon commonly used in sensors of this type, light can be delivered in a very simple and compact way to probe the change in the sensor's properties to determine whether or not any molecules of interest have been captured.
|
|
"If you look at a CD or a DVD in the light, you'll notice lots of colors bounce off it in room light. The way these discs work is that there are a bunch of bumps, which are very similar to our gratings," says Weiss. When light interacts with a particular arrangement of bumps, the engineers can essentially tell the light what to do, enabling readout of the music or videos on the disc.
|
|
That's precisely what the Vanderbilt team has done with their sensor—put bumps on top of it to control how the light interacts with their active sensing medium (the porous silicon "waveguide"). Then they can determine what the signal means based either on the color that comes out or by the angle of light as it exits the sensor.
|
|
"When we infiltrate the molecules that we want to detect and they stay in the sensor and attach, they change the optical density of the porous silicon and, consequently, the angle or the color of light that comes back out," Weiss elaborates. "By knowing how much the angle changes, for example, we can quantify how many molecules are present. So not only can we identify our DNA sequence or toxin, but we can also know how much is present as well. For diagnostics, it's very helpful to know how much is present."
|
|
Other potential applications beyond DNA sensing may include detecting small molecules—such as toxins in the environment—which has significant national security implications.
|

