| Posted: July 18, 2007 |
Nanoparticles send peptides round the twist |
|
(Nanowerk News) Nanoparticles with flexible side chains cause peptides to adopt a helical form, making them promising anticancer agents.
|
|
Vincent Rotello and colleagues at the University of Massachusetts, Amherst, US, used functionalised gold nanoparticles to persuade a tetraaspartate peptide (TAP) to form an alpha-helix. In the absence of the nanoparticles, the peptide forms a random coil ("Binding and templation of nanoparticle receptors to peptide -helices through surface recognition" - free access article).
|
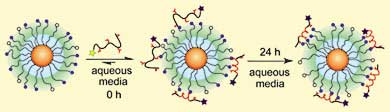 |
Ionic interactions between the ligands on the nanoparticle's surface, and the peptide in solution, cause the peptide to form a helix.
|
|
In preliminary studies, Rotello and colleagues had used trimethylammonium functionalised gold nanoparticles to stabilise TAP into an alpha-helix. However, these particles were not biocompatible, limiting their potential applications. Now they have overcome this problem by using tetra(ethyl glycol) (TEG) ligands, which are biocompatible.
|
|
Lucia Pasquato, an expert in functional nanoparticles at the University of Trieste, Italy, explained that specific groups present on the surface of nanoparticles can induce a peptide to assume a particular conformation by ionic interactions.
|
|
Alpha-helices, common protein secondary structures, are involved in many protein-protein interactions. Some of these interactions take place in pathways critical in certain human diseases, such as cancer. By mimicking the alpha-helix of relevant proteins, it may be possible to create new cancer therapies.
|
|
Pasquato added, 'this is a first step towards the designing of more sophisticated artificial systems able to compete with nature in performing multivalent interactions for recognition processes.'
|

