| Aug 20, 2011 |
Seeing the world of nanotechnology from a single-molecule perspective
|
|
(Nanowerk News) Observing the structure of collapsing unstable atomic nuclei using electrons is an experimental goal that has not been achieved anywhere in the world. Masanori Wakasugi, director of the Instrumentation Development Group at the RIKEN Nishina Center for Accelerator-Based Science (RNC), is working on this challenging issue. The current theoretical model of the atomic nucleus has been constructed with major contributions from electron-scattering experiments, in which electrons are collided with stable atomic nuclei to visualize the nuclear structure. In recent years, however, a wide range of experiments on the properties of unstable atomic nuclei has revealed a number of phenomena that are inconsistent with the current model of the atomic nucleus. Radioisotope–electron scattering experiments in which electrons collide with unstable nuclei are indispensible in establishing the ultimate model of the atomic nucleus, which will yield a comprehensive understanding of both stable and unstable nuclei. Wakasugi and his colleagues are taking unique approaches to achieve this world-first experiment.
|
|
Observing the chemical reactions of single molecules
|
|
"When I was in junior high school, I learned the chemical formula for the electrolysis of water," says Kim. That formula is H2O → H2 + 1/2O2. "I asked my teacher why we need to multiply the O2 by half. The teacher answered that the oxygen is multiplied by half because when water is electrolyzed, hydrogen and oxygen are produced in the proportion of two to one. However, I thought, what if a single water molecule is electrolyzed? This question gave me the incentive to observe the process of a chemical reaction on the scale of a single molecule."
|
|
Kim went on to the Department of Chemistry at Seoul National University where he majored in electrochemistry. "At that time, I conducted experiments that used an electrical circuit, like in the electrolysis of water, to control a chemical reaction in a solution and to examine the chemical reaction from the reaction products. This approach, however, does not provide information on how individual molecules are involved in a chemical reaction. We can only conjecture."
|
|
After finishing his master degree program at Seoul National University, he visited Japan in 1996 and started research at The University of Tokyo under the supervision of Akira Fujishima, now president of the Tokyo University of Science, who was known as the 'father of the photocatalyst'. Photocatalysis is a process by which molecules can be broken down on the surface of a photoactive material, such as titanium oxide, on exposure to light. "I originally planned to make a thorough study of photocatalysts. However, Prof. Fujishima suggested that I do more basic research because my background was in science. So I decided to study the physical phenomena that occur when the surface of a substance is exposed to light."
|
|
Reacting a single molecule
|
|
"When I was in the third year of my doctoral program, I came across a very intriguing paper reporting that a scanning tunneling microscope had been successfully used to observe the 'molecular vibration' of a single molecule. I immediately thought that this was what I really wanted to do."
|
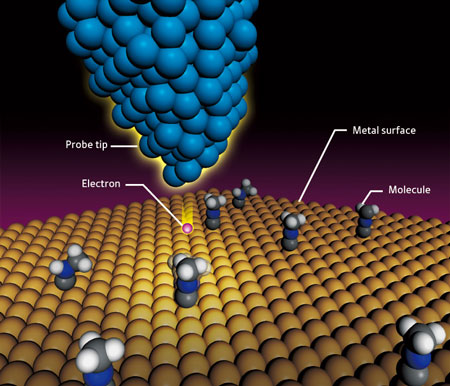 |
| Figure 1: The principle of scanning tunneling microscopy. When a voltage is applied to an atomically sharp STM tip that is brought close to a molecule on a metal surface, a tunneling current flows between the tip and the molecule, injecting electrons into the molecule and inducing a molecular vibration. The intensity of the molecular vibration at a given voltage can be used to identify the molecule. This technique can also be used to induce a chemical reaction.
|
|
A scanning tunneling microscope (STM) is an imaging technique that allows the microscopic surface structure of a substance to be mapped at resolutions approaching the scale of individual atoms. But this is not the only function of STM; it can also be used to identify the types of molecules present based on the molecular vibration.
|
|
In STM, a voltage is applied to a very sharp probe tip that is brought very close to a molecule on a surface (Fig. 1). Electrons from the probe flow to the target molecule, producing what is called a 'tunneling current', referring to the way electrons seem to 'tunnel' through the classical energy barrier needed for such a current to flow. This current induces a molecular vibration, causing all the individual atoms of the target molecule to become displaced from their equilibrium positions. The intensity of the molecular vibration corresponding to a given voltage depends on the type of molecule or the chemical bonds within the molecule. The type of molecule can therefore be identified by observing the molecular vibration.
|
|
"I was searching for a research laboratory where I could use STM in Japan when Prof. Fujishima introduced me to the Surface Chemistry Laboratory at RIKEN, headed at that time by Chief Scientist Maki Kawai, who is now an Executive Director of RIKEN."
|
 |
| Figure 2: Yousoo Kim adjusting a scanning tunneling microscope.
|
|
After joining the Surface Chemistry Laboratory in 1999, Kim developed STM technologies together with Tadahiro Komeda, a research scientist in the laboratory and now a professor at Tohoku University. There, Kim observed molecular vibrations to successfully identify individual molecules on this basis (Fig. 2). He also succeeded in injecting electrons into a specific site of a molecule, thus changing it into a different molecule (Fig. 3).
|
|
"We removed two hydrogen atoms from a trans-2-butene molecule consisting of four carbon and eight hydrogen atoms to produce a 1,3-butadiene molecule consisting of four carbon and six hydrogen atoms. We used STM to cause a chemical reaction as intended within a single molecule, observed the vibrational signals before and after the reaction, and identified the type of molecule successfully for the first time."
|
|
Kim attributes the success in eliciting the desired chemical reaction to the laboratory's earlier work in catalysis. "We placed a molecule on the surface of palladium, which served as a catalyst for the chemical reaction. The Surface Chemistry Laboratory originally started as a catalyst research laboratory, and we owe much to the huge accumulation of knowledge on molecules and catalysts on the surface of substances."
|
|
Controlling individual molecules
|
|
There still remained a technical challenge to be overcome in observing molecular vibrations by STM. "When electrons are injected from an STM probe tip into a molecule, some molecules start moving before their molecular vibrations are observed. Finding an effective way to observe these unstable molecules was a big problem for us."
|
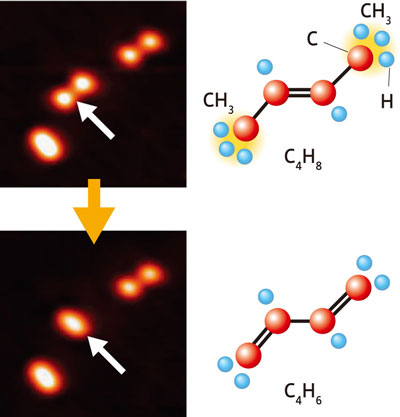 |
| Figure 3: Chemical reactions on a single-molecular level. Electrons were injected from an STM tip into the two methyl groups (CH3) of a trans-2-butene (C4H8) molecule (upper), which released a single hydrogen atom (H) from each of the methyl groups and turned into a 1,3-butadiene (C4H6) molecule (lower). Arrows point to the molecule that caused the chemical reaction.
|
|
Kim and his laboratory colleagues examined what electron energy level causes the molecule to move. "As a result, we found that the molecule moves at an injected electron energy level equal to that causing the strongest molecular vibration." Based on these experiments, they established a unique measurement method called 'action spectroscopy'. "This measurement method made it possible for us to identify all types of molecules, both stable and unstable molecules, and to examine their essential characteristics."
|
|
When electrons are injected from an STM probe tip into a molecule, the molecule can move in many directions. "We cannot control the direction of a molecule's movement, but we encounter this problem only when the STM probe tip is placed right above the molecule. So we placed the STM probe tip obliquely upward and used the electrostatic force acting between the probe tip and the molecule. This approach also enabled us to control the direction of movement of the molecule successfully."
|
|
Kim's team has used this technique to draw letters by moving molecules (Fig. 4). In the late 1980s, a paper was published describing an experiment in which the atoms forming a molecule were moved by STM to construct letters. In that experiment, the letters were created by drawing the atoms closer to the probe tip or by using the tip to shape the atoms. "We constructed our letters by moving the molecules themselves in the desired direction on a surface. This cannot be achieved without a complete understanding of the nature of molecules and the interaction between electrons and molecules." In the future, this technique will be applied in the fabrication of computer circuits by arranging molecules.
|
|
Electrolyzing single water molecules
|
|
In 2009, Kim started the experiment that he first imagined when he was in junior high school—the experiment to electrolyze a single water molecule. "In electrolyzing a single water molecule, there are two possible reaction pathways," he says. Those pathways are H2O → 2H + O, and H2O → H + OH. In the former reaction, the two hydrogen atoms are separated from the single oxygen atom, and can be achieved by injecting electrons with high energy. The difficulty is how to produce the other reaction pathway.
|
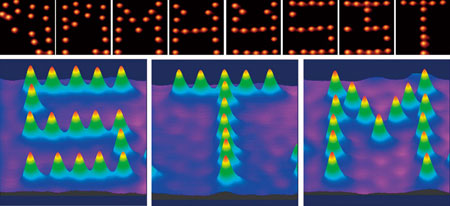 |
| Figure 4: Letters drawn using an STM tip to move molecules. Electrostatic force between organic molecules (CH3S) and the STM tip was used to move the organic molecules to form the letters S, T and M (lower). The upper pictures show the drawing process for each letter.
|
|
Electrons injected into a molecule from an STM tip cause the molecule to start vibrating in an excited state. If the duration of the excited state (vibrational lifetime) is long enough, the molecular vibration causes the bonds between the atoms to break down, which increases the probability of a chemical reaction occurring. "When a single water molecule is placed on the surface of a metal, the water molecule cannot be broken down because of its short vibrational lifetime. This is because the water molecule binds chemically to the metal surface, and the energy of the injected electrons is easily dissipated into the metal surface."
|
|
Placing a water molecule on the surface of an insulator instead of a metal can increase the vibrational lifetime because no chemical reactions can occur and no electronic energy is absorbed. However, a tunneling current cannot flow from the STM probe tip in this case because the water molecule is on an insulator. "To cope with this problem, we developed a metal surface coated with an ultrathin film of magnesium oxide just two atoms thick. A water molecule on this surface produces a small tunneling current in STM."
|
|
Theoretically, a water molecule can be electrolyzed when injected with an electron having an energy of 0.77 electronvolts or more. On the ultrathin MgO film, however, the water molecule broke down at just 0.45 electronvolts. "We attributed this to a multi-step excitation process in which the water molecule is excited by the first injected electron and then by the following injected electron while the water molecule is still in the vibrationally excited state, because the electron energy is slowly dissipated owing to the ultrathin insulating film surface and hence the vibrational lifetime is increased."
|
|
The results of their experiments showed exactly what they were looking for (Fig. 5). "Using this approach, we succeeded in separating a single hydrogen atom from a single water molecule," says Kim. These results confirmed the H2O → H + OH reaction pathway experimentally for the first time, and could lead to the development of technologies for producing hydrogen fuel with the minimum consumption of energy.
|
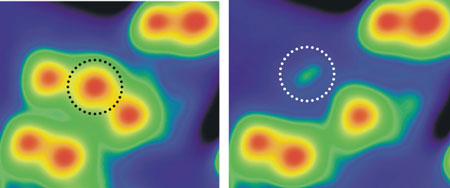 |
| Figure 5: Electrolysis of a single water molecule by a new reaction pathway. Electrons were injected from an STM tip into a single water molecule on the surface of an MgO film (at the point indicated by a circle in the picture on the left) to cause the water molecule to vibrate. The single water molecule was successfully electrolyzed in this way according to the reaction pathway: H2O → H + OH. The circle in the picture on the right indicates an OH group as an electrolyzed product.
|
|
Practical applications of single-molecule experiments
|
|
In 2010, Kim started the Surface and Interface Science Laboratory at the RIKEN Advanced Science Institute. "We are working on new research into the interaction between light and substances. Many researchers have already investigated this subject. However, there have been virtually no reports on experiments that examine the interaction between light and substances while observing individual molecules."
|
|
Photocatalysts are a firm research target. "In Prof. Fujishima's laboratory, I used to watch how he advanced his own research into photocatalysts around him. This time, I intend to conduct research into the essence of photocatalysts in my own right based on the technology and experience I gained over the years at RIKEN."
|
|
On a single-molecular scale, nobody knew the position on titanium oxide at which a photocatalytic reaction occurs. "It has been considered for years that the photocatalytic reaction occurs at positions where oxygen atoms are missing on the surface of titanium oxide because electrons concentrate at those positions. Our experiments with an STM probe tip clarified that photocatalytic reactions actually occur across wide electronically active areas around the positions where oxygen atoms are missing."
|
|
The Surface and Interface Science Laboratory is also conducting research into organic solar cells. "What types of molecules are most effective and how should we arrange them to increase power generation efficiency? Many researchers from around the world have wanted to perform single-molecule experiments while observing individual molecules, but such experiments have been too difficult to handle. We have accumulated STM technology that I am confident will enable such experiments."
|
|
Toward 'sci-engineering'
|
|
"So far, I have focused on research into the essence of chemistry. In the future I also plan to start research that helps us link that knowledge to practical applications. This idea was triggered by a meeting with Dr Takanori Fukushima from the Energy Conversion Research Team. He specializes in organic synthesis and can synthesize any organic molecule. I always have a good time with him, talking about our dreams."
|
|
Molecules and matter exhibit different characteristics on the nanometer or molecular scale compared with the macroscale behavior scientists are most familiar with. This is the reason for the widespread scientific interest in nanotechnology over the past ten years, and the origin of the expectations for a nanotechnology revolution.
|
|
"These expectations, however, are now on the point of fading because the findings to date have fallen short of society's expectations. Although many theoretical papers have been published on what is actually going on in the nanometer world, only a few study have been reported because of the technical difficulty in directly observing the nature and functions of individual molecules. Many conventional application studies have been conducted without fully understanding the basic mechanisms of nanotechnology. I plan to make use of the STM to study the nature of individual molecules and open a new frontier in nanoscience that will allow us to explore the essence of the nanoworld.
|
|
"RIKEN launched systematic research into nanoscience before anywhere else in the world," Kim points out. "In 1993, Dr Kawai, now an Executive Director of RIKEN, started the Atomic Scale Sci-engineering Research and Promotion Group together with Chief Scientist Masakazu Aono, now a fellow at the National Institute for Materials Science, and Chief Scientist Katsunobu Aoyagi, who is now professor at Ritsumeikan University. 'Sci-engineering' is a term implying that research into the essence of a phenomenon should come first, and then engineering should follow from the results. I would like to follow the research concept of sci-engineering in the Surface and Interface Science Laboratory.
|





