| Oct 31, 2011 |
Solar cells: The dyes have it
|
|
(Nanowerk News) The dye-sensitized solar cell (DSC) is a next-generation photovoltaic technology with the potential to reduce the cost of solar energy production to levels comparable to that of fossil fuel-based electricity generation. DSCs use a mix of dye molecules and inexpensive titanium dioxide (TiO2) nanoparticles to convert sunlight into electricity, and can be easily and cheaply fabricated into thin, large-area sheets using simple screen-printing techniques. Despite the promise of this technology, however, achieving the high conversion efficiencies needed to approach commercial viability has meant using resource-limited materials such as ruthenium.
|
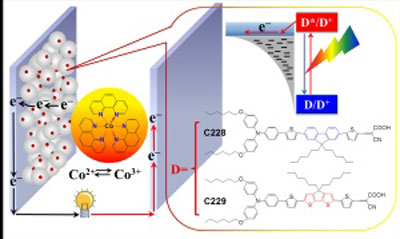 |
| The push–pull dye C229 is coupled with a cobalt-based electrolyte to achieve high solar energy conversion efficiency.
|
|
Peng Wang, Yu Bai and colleagues from the Chinese Academy of Sciences in Changchun have now found a way to achieve high DSC conversion efficiency without ruthenium by developing an all-organic dye that can be used with more readily available cobalt ("Engineering Organic Sensitizers for Iodine-Free Dye-Sensitized Solar Cells: Red-Shifted Current Response Concomitant with Attenuated Charge Recombination").
|
|
The best-performing DSCs at present use ruthenium dyes, coupled with an iodine-based electrolyte, to harvest light and transfer photogenerated free electrons into a surrounding mesoporous matrix of TiO2. The high price and restricted availability of ruthenium has led researchers to pursue alternative 'push–pull' dye sensitizers — organic molecules containing electron-accepting and -donating groups linked together by a conjugated bridge. Finding a suitable organic dye, however, has been a challenge.
|
|
Wang and his co-workers improved on previous strategies by incorporating an aromatic–sulfur bridging group into the push–pull dye C229. This modification enhanced the dye's ability to collect red light from the visible spectrum, allowing it to absorb more solar light overall (see image). Combining this photo-sensitizer with cobalt phenanthroline as an electrolyte to regenerate the dye resulted in a DSC system with an energy conversion efficiency of 9.4% under typical daylight conditions — a new benchmark value for ruthenium-free DSCs and only marginally behind the best ruthenium-based systems. The use of a cobalt-based electrolyte also addresses another shortcoming of ruthenium-based DSCs: the need for an iodine electrolyte that is highly corrosive and reduces cell voltages.
|
|
Measurements and quantum calculations revealed that interactions between the cobalt electrolyte and the C229 dye helped lift the system's reduction–oxidation potential above its ground state, making the cell more receptive to light across the solar spectrum. "This observation is very encouraging for developing dyes with smaller energy gaps," notes Bai.
|

