| Posted: November 28, 2007 |
Mastering molecular memory |
|
(Nanowerk News) A new molecule that switches shape when triggered by light could lead to nanoscale memory devices, say chemists in Japan ("Reversible photochromism of a ferrocenylazobenzene monolayer controllable by a single green light source" – free access article).
|
|
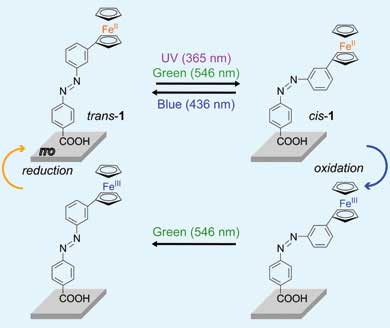
Hiroshi Nishihara and colleagues from the University of Tokyo have combined photochemistry and electrochemistry to make a molecule that can be switched from one form to another, and then back again, using a single source of light. Previous such photochromic molecules have needed a second light source of a different wavelength to be flipped back to their original state.
|
 |
|
'Focussing dual light sources on the same small spot can be a problem from the perspective of both technical difficulty and cost performance,' said Nishihara. 'Our system is the first monolayer film of photochromic molecules which can be reversibly switched by a single light source.'
|
|
The Tokyo team used a photochromic molecule that incorporates an iron-containing ferrocene group, which they deposited in a single layer onto a transparent electrode surface. When the molecule's iron core is in the 2+ oxidation state, green light isomerises a double bond in the structure from trans to cis, changing the shape. When the iron is electrochemically converted to Fe(III), the same green light will convert cis form back to the trans isomer.
|
|
'This work takes one of the most challenging leaps for chemists: to interface small molecule materials with developing infrastructure from microtechnology,' said Amar Flood, who researches molecular switches at Indiana University, Bloomington, US. 'It is critical to take this step - chemists mustn't lose sight of engineering, because otherwise our engineering colleagues will lose sight of us.'
|
|
Nishihara is now working to fine-tune the chemical structure of the molecule, to increase the proportion of molecules in the sample that isomerise in response to the light. 'The other [project] is to immobilise the molecules onto submicron-sized electron arrays, to demonstrate high density memory device fabrication,' he said.
|

