| Posted: December 14, 2007 |
Enzyme-powered delivery vehicles |
|
(Nanowerk News) Dutch scientists have made nanotubes move using enzyme-powered motors.
|
|
Ben Feringa and co-workers from the University of Groningen, The Netherlands, have designed engines for nanomachines that could potentially be used in the body ("Autonomous propulsion of carbon nanotubes powered by a multienzyme ensemble" - free access article).
|
|
Hydrogen peroxide has proven useful as a chemical fuel for powering microscopic motors but its practicality is somewhat limited by its inherent reactivity, said Feringa. To get around this problem the team have used two enzymes in tandem as the engine for their nanomachine. They explained that by coupling glucose oxidase with catalase, relatively stable glucose can be used as the primary fuel instead. 'This fuel is already present in the body,' said team-member Wesley Browne, 'and it is completely inert.'
|
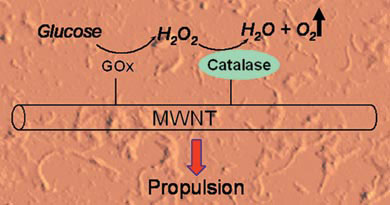 |
| Oxygen gas produced by enzyme reactions causes the nanotube to move
|
|
The glucose oxidase converts glucose and oxygen to gluconolactone and hydrogen peroxide. The hydrogen peroxide is then consumed by the catalase to produce water and oxygen. The results are surprising, said the team, because in principle nothing should happen - more oxygen is consumed than is produced. But the glucose oxidase produces high local concentrations of hydrogen peroxide, which in turn results in bursts of oxygen being released as bubbles of dioxygen gas. This is what causes the enzymes (and the nanotubes they are attached to) to move.
|
|
'This work is the first step towards the design of functional nano-machinery working with renewable resources as fuel,' said Heinz-Bernhard Kraatz, an expert on nano-biomaterials from the University of Western Ontario, London, Canada.
|
|
While the nanotubes' movement currently appears erratic, the Dutch team have noticed it is dependent on the shape of the nanotube/enzyme aggregate and the enzymes' position on the nanotube. Although currently only non-directional movement is demonstrated, said Kraatz, 'it is clear that the system can be upgraded as soon as rational and site directed modifications of nanosized objects becomes available.'
|

