| Posted: March 28, 2008 |
Strength in nanoworms |
|
(Nanowerk News) Scientists in the US have mimicked the structure of bird nests to overcome the extreme fragility of highly insulating aerogels.
|
|
Already used to protect NASA's Mars rovers' delicate electronics from the cold, aerogels are low-density materials prized for their insulating properties. However, the porous materials, made of interwoven nanoparticle strands, are also extremely fragile, limiting their use. Now Nicholas Leventis and colleagues at Missouri University of Science and Technology, Rolla, have made a stronger material by boosting the interlocking between strands ("Polymer nanoencapsulated mesoporous vanadia with unusual ductility at cryogenic temperatures" – free access article).
|
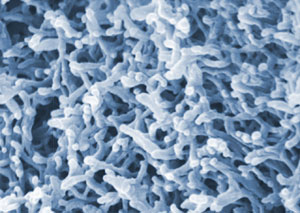 |
| Inspired by the highly interlocked structures of bird nests, vanadia-based structures which form a more highly entangled worm-like nanostructure were made.
|
|
Aerogels are typically made from pearl necklace-like strings of silica nanoparticles, and can be strengthened with a polymer coating, so that the strands form crosslinks wherever they meet. But inspired by the highly interlocked structures of bird nests, Leventis switched to vanadia-based structures, which form a more highly entangled worm-like nanostructure.
|
|
'Both crosslinked silica and vanadia are very strong materials. But crosslinked vanadia aerogels never fail under compression, and can absorb at least four times the kinetic energy of the silicon carbide ceramics used for armour,' said Leventis. 'Killer applications will be in areas where we can take advantage of the multifunctional character of these materials - strength in combination with acoustic and thermal insulation - such as lightweight structural materials for buildings, and the automotive and airplane industries.'
|
|
Bakul Dave who makes similar gel-based materials at Southern Illinois University, Carbondale, US, describes the work as a step forward. 'It seems the properties depend on the nanostructure morphology - so in principle, it should be possible to make these materials with components other than vanadium,' says Dave.
|
|
Replacing vanadium with a cheaper alternative is Leventis' first goal. 'We are applying lessons learned from vanadia to silica, which under certain conditions can also produce worm-like structures,' Leventis adds.
|

