| Posted: June 27, 2008 |
Installing 'traffic lights' inside living cells |
|
(Nanowerk News) Cells live their days on the fixed schedule of the cell cycle, which regulates when—and if—they divide. Some cells cycle rapidly and, as a result, divide actively. For others, the cell cycle is stalled; these will not divide, but may instead differentiate into mature cells that no longer reproduce.
|
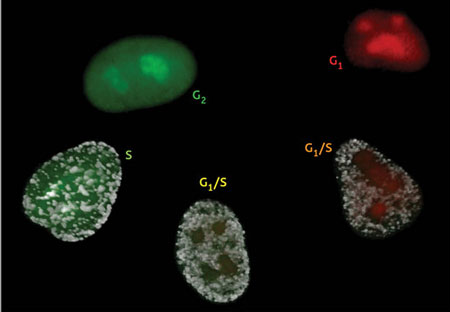 |
| The Fucci cell cycle visualization method. Cells in phase G1 fluoresce bright red. As they transition into the S phase and begin DNA replication, Cdt1 levels are dramatically reduced while Geminin levels increase. This results in faint yellow fluorescence early in G1/S that soon gives way to robust green fluorescence, which lasts until the cell re-enters G1 phase. (Image: RIKEN)
|
|
Understanding cell cycle behavior of individual cells in complex tissues would be valuable in studying processes like embryonic development and tumor formation, but this has proven difficult. “Considerable progress has been made towards understanding the mechanism of cell cycle progression in single cells,” explains Atsushi Miyawaki of the RIKEN Brain Science Institute in Wako. “However, little is known about how the cell cycle is coordinated with differentiation, morphogenesis, and cell death in a multicellular context.”
|
|
To ensure tight regulation of cell division, cells must maintain close control over levels of proteins involved in the cell cycle. For example, the proteins Cdt1 and Geminin regulate chromosomal DNA replication during the ‘S’ phase of the cell cycle. These molecules have opposing effects on DNA replication, and cells control their relative levels via timed, targeted destruction of the unwanted protein. Directly before DNA replication, in the ‘G1’ phase, Cdt1 levels are highest; as cells transition into S phase, Cdt1 levels fall and Geminin levels rise, remaining high until the cell returns to G1.
|
|
Miyawaki and colleague Asako Sakaue-Sawano took advantage of this closely regulated process to develop a surprisingly precise visual indicator of cell cycle status in living cells ("Visualizing Spatiotemporal Dynamics of Multicellular Cell-Cycle Progression"). In their technique, called ‘Fucci’, cells are genetically modified to express Cdt1 and Geminin with red and green fluorescent tags, respectively. As a result, actively replicating cells in S phase glow green, while cells that are in G1 and not yet actively dividing will glow red; cells in transition glow yellow, as their DNA begins to replicate.
|
|
After injecting Fucci-expressing tumorigenic cells into mice, Miyawaki’s team was able to directly observe active tumor cell division in living animals over the course of a month. For subsequent experiments, the group generated transgenic Fucci-expressing mice, in which they were able to perform exquisitely detailed analyses of cell cycle profiles of cells in various regions of the developing brain.
|
|
These are strong demonstrations of Fucci’s capabilities, but Miyawaki believes this is only the beginning: “Future challenges involve further developing Fucci derivatives with different colors … that highlight cell-cycle transitions other than G1/S, and that can function in non-mammalian cell types.”
|

