| Jul 02, 2013 |
New method for mapping the protein signals between healthy and diseased cells
|
|
(Nanowerk News) Researchers at Memorial Sloan-Kettering Cancer Center in New York, working in collaboration with researchers at the Proteome Center Tuebingen (PCT), have developed a new method for identifying the cell of origin of intracellular and secreted proteins within multicellular environments ("Cell-selective labeling using amino acid precursors for proteomic studies of multicellular environments").
|
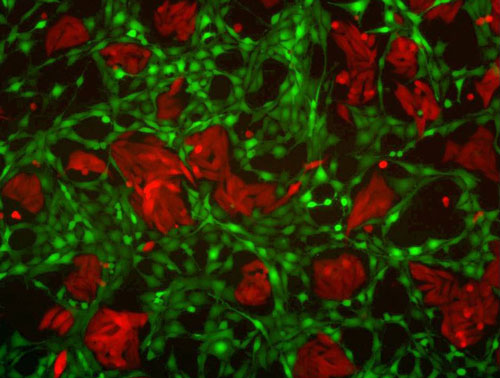 |
| Microscope image of human breast cancer cells (red) grown together with mouse fibroblasts (green). This mixed-species culture was used to validate the ability of the CTAP methodology to distinguish proteins from each cell population. (Image: Nicholas Gauthier, Chris Sander, Martin Miller)
|
|
This technological advancement is particularly exciting because it will provide investigators with a new tool for comprehensive mapping of cell-cell communication, which is especially important in all aspects of cancer development, maintenance, and response to therapy. For example, this method could be used to study cell signaling events between normal and malignant cells in order to better understand the molecular mechanisms by which surrounding normal cells alter tumor growth and response to treatment.
|
|
The technique, named cell type specific labeling using amino acid precursors (CTAP), exploits the inability of vertebrate cells to synthesize essential amino acids normally required for growth and homeostasis. A team headed by Dr. Nicholas Gauthier and Dr. Martin Miller at the Memorial Sloan-Kettering Cancer Center engineered cells to express amino acid biosynthesis enzymes, which enabled cells to grow on their own supply of amino acids produced from supplemented precursors.
|
|
The team went on to show that supplementing heavy stable isotope-labeled forms of these precursors led to incorporation of heavy amino acids into proteins produced in enzyme expressing cells. Dr. Boumediene Soufi and Dr. Boris Macek from the PCT designed experiments that utilized quantitative mass spectrometry to search for proteins that contained these stable isotope labels. In this way, the cell of origin of both intracellular and secreted proteins identified in multicellular culture could be determined. By providing a means to link proteins directly to specific cell types, the authors believe that this new method will be useful in studies of cell-cell communication and biomarker discovery.
|

