| Jul 31, 2013 |
3D molecular syringes
|
|
(Nanowerk News) Abdominal pain, fever, diarrhoea – these symptoms could point to an infection with the bacterium Yersinia. The bacterium’s pathogenic potential is based on a syringe-like injection apparatus called injectisome. For the first time, an international team of researchers including scientists at the Helmholtz Centre for Infection Research (HZI) in Braunschweig, Germany, has unraveled this molecular syringe’s spatial conformation. The researchers were able to demonstrate that the length of Yersinia’s injectisome’s basal body, which crosses the bacterial cell wall, is adjustable – very likely an adaptation to physical stress.
|
|
The rod-shaped bacterium Yersinia enterocolitica, which is transmitted through contaminated food, causes gastrointestinal diseases. In Germany alone, several thousand cases are reported annually. Yersinia uses a rather sophisticated tool – its injection apparatus – to infect humans. Not only does the apparatus look like a syringe, it actually serves a similar purpose. A molecular “needle”, which sticks out from the bacterium’s surface, extends across the bacterial membranes to the host cell. It is through this needle that the bacterium “injects” substances that facilitate infection of the host. Now, for the first time, an interdisciplinary team of HZI scientists together with their colleagues at the Biozentrum of the University of Basel and at the Ecole Polytechnique Fédérale de Lausanne in Switzerland, has presented the structure of Yersinia enterocolitica‘s injectisome in high-resolution and 3D. They published their results in the digital scientific magazine eLife ("In situ structural analysis of the Yersinia enterocolitica injectisome").
|
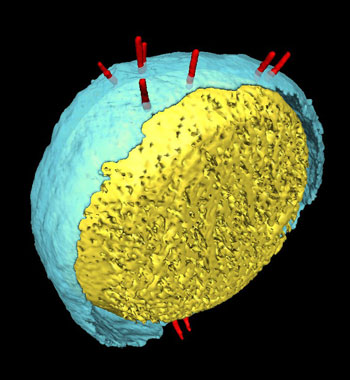 |
| Electron microscope image of a bacterial cell: injection apparatuses (shown in red) stick out from the cell and extend across the cell wall (shown here in yellow and blue).
|
|
Their innovative approach has yielded surprising results. Previous studies had been concerned with isolating the molecular syringe from the bacterium and studying it under the electron microscope. “We, however, actually studied the injectisome in situ, in other words, on the bacterial surface, right where it normally occurs,” explains Prof. Henning Stahlberg, University of Basel. To this end, the researchers cooled the bacteria to minus 193 degrees Celsius and used cryo-electron microscopy to take pictures of the syringe from various angles. They then computed a spatial structure from a set of two-dimensional images – a highly effective method for examining large molecular complexes. The syringe, which consists of some 30 different proteins, definitely falls into that category.
|
|
When comparing over 2000 single syringes from over 300 bacteria, the researchers made a surprising discovery: “There is a range of different lengths of each injection apparatus’ base – in some cases, it’s on the order of ten nanometers, or ten millionth of a millimeter. It can be stretched or compressed – just like a spring,” explains Dr. Stefan Schmelz of the HZI, one of the study’s first authors. As much as we consider such dimensions to be miniscule – to a bacterium, which itself is but a hundred times that size, they are substantial. “Bacteria are exposed to considerable forces, be it during contact with other cells or upon changes in environmental salinity,” explains Prof. Dirk Heinz, the HZI’s scientific director and former head of the HZI Department of Molecular Structural Biology. “If the injectisomes were rigidly constructed, bacteria would most likely be unable to resist these forces. Their cell walls would simply rupture.”
|
|
Insights into the structure of Yersinia’s attack tool offer clues as to ways in which the molecular syringe may be therapeutically inhibited. Without this apparatus, the bacteria are practically harmless. “Also other pathogenic bacteria make use of this principle during infection, for example Salmonella that cause food poisoning,” confirms Dr. Mikhail Kudryashev, another of the study’s primary authors and a researcher at the University of Basel. The team was already able to document this same flexibility in Shigella, the causative agent behind bacillary dysentery. The “molecular building kit,” as Schmelz calls it, is highly similar, suggesting that insights from this current study can potentially also be applied to other pathogenic bacteria.
|

