| Apr 16, 2014 |
At the origin of cell division
|
|
(Nanowerk News) Droplets of filamentous material enclosed in a lipid membrane: these are the models of a “simplified” cell used by the SISSA physicists Luca Giomi and Antonio DeSimone, who simulated the spontaneous emergence of cell motility and division - that is, features of living material - in inanimate “objects”. The research is one of the cover stories of the April 10th online issue of the journal Physical Review Letters ("Spontaneous Division and Motility in Active Nematic Droplets").
|
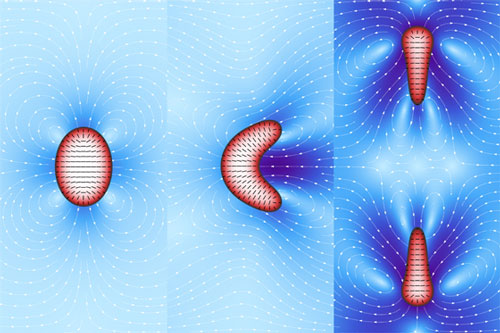 |
| active droplets
|
|
Giomi and DeSimone’s artificial cells are in fact computer models that mimic some of the physical properties of the materials making up the inner content and outer membrane of cells.
|
|
The two researchers varied some of the parameters of the materials, recording what happened: “our ‘cells’ are a ‘bare bones’ representation of a biological cell, which normally contains microtubules, elongated proteins enclosed in an essentially lipid cell membrane”, explains Giomi, first author of the study. “The filaments contained in the ‘cytoplasm’ of our cells slide over one another exerting a force that we can control”.
|
|
The force exerted by the filaments is the variable that competes with another force, the surface tension that keeps the membrane surrounding the droplet from collapsing. The generates a flow in the fluid surrounding the droplet, which in turn is propelled by such self-generated flow. When the flow becomes very strong, the droplet deforms to the point of dividing. “When the force of the flow prevails over the force that keeps the membrane together we have cellular division”, explains DeSimone, director of the SISSA mathLab, SISSA’s mathematical modelling and scientific computing laboratory.
|
|
“We showed that by acting on a single physical parameter in a very simple model we can reproduce similar effects to those obtained with experimental observations” continues DeSimone. Empirical observations on microtubule specimens have shown that these also move outside the cell environment, in a manner proportional to the energy they have (derived from ATP, the cell “fuel”). “Similarly, our droplets, fuelled by their ‘inner’ energy alone – without forces acting from the outside – are able to move and even divide”.
|
|
More in detail ...
|
|
“Acquiring motility and the ability to divide is a fundamental step for life and, according to our simulations, the laws governing these phenomena could be very simple. Observations like ours can prepare the way for the creation of functioning artificial cells, and not only”, comments Giomi. “Our work is also useful for understanding the transition from non-living to living matter on our planet. The development of the early forms of life, in other words.”
|
|
Chemists and biologists who study the origin of life don’t have access to cells that are sufficiently simple to be observed directly. “Even the simplest organism existing today has undergone billions of years of evolution”, explains Giomi, “and will always contain fairly complex structures. Starting from schematic organisms as we do is like turning the clock back to when the first rudimentary living beings made their first appearance. We are currently starting studies to understand how cell metabolism emerged”.
|

