| Sep 25, 2014 |
Unlocking long-hidden mechanisms of plant cell division
|
|
(Nanowerk News) Along with copying and splitting DNA during division, cells must have a way to break safely into two viable daughter cells, a process called cytokinesis. But the molecular basis of how plant cells accomplish this without mistakes has been unclear for many years.
|
|
In a new paper by cell biologist Magdalena Bezanilla of the University of Massachusetts Amherst, she and her doctoral student Shu-Zon Wu present a detailed new model that for the first time proposes how plant cells precisely position a “dynamic and complex” structure called a phragmoplast at the cell center during every division and how it directs cytokinesis. The work is reported in the current issue of the journal, eLife ("Myosin VIII associates with microtubule ends and together with actin plays a role in guiding plant cell division").
|
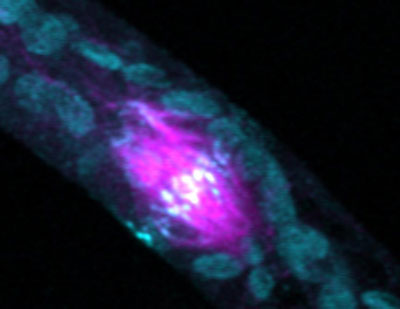 |
| A phragmoplast with microtubules in magenta and myosin VIII in cyan blue. The large globular objects in cyan blue are chloroplasts, which auto-fluoresce. There is no myosin VIII on those structures. A new model of how plant cells position phragmoplasts during cell division to direct cytokinesis answers what has been an open question for decades in cell biology.
|
|
The complicated cytokinesis process begins at the cell center where the phragmoplast sends out tendrils toward opposite sides of the cell like a belt across its waist. These polymers, microtubules and actin filaments travel tens or hundreds of microns towards the cell wall to a predetermined position where a special structure called the cell plate will form. The new wall making two new cells will take shape along this pathway between the cell plates on opposite walls.
|
|
“How this process is directed and accomplished has been a mystery for a very long time,” says Bezanilla. But clearly, the scientist adds, microtubules are needed here because without them the cell plate does not form. “And we’ve known for a very long time that actin filaments are there in the structure, but nobody knew what they were for. What steers phragmoplast expansion at the molecular level was just not understood.”
|
|
“Our new paper proposes a model showing how plant cells steer their cell division machinery into position and how actin contributes. Our data from experiments in moss and tobacco provide evidence that a protein called myosin VIII, along with actin, guide cytokinesis. It answers what has been an open question in cell biology for decades.”
|
|
Using a state-of-the-art microscope funded by the Massachusetts Life Sciences Institute at UMass Amherst, she and Wu were able to watch key structures taking shape, label them and make videos of cytokinesis for hours to piece together how actin, microtubules and the structural protein known as myosin VIII cooperate to accomplish proper division.
|
|
“What’s unique with this myosin is that it can also interact with microtubules and in fact we think the microtubules are its cargo,” Bezanilla says. “We think the myosin is pulling the microtubule along an actin filament.” Crosstalk between actin and microtubules is something that happens in all cells so this work in plants could have implications in animal cell processes as well, Bezanilla says.
|
|
She adds, “Some of these things were just at the edge of being visible. It was quite a feat to be able to image the process for such a long time and to witness a live cell process.”
|
|
Overall, using a combination of genetics and live-cell imaging to query what guides the phragmoplast, the scientists identified actin and actin-based molecular motors, the class VIII myosins, as a key to the steering mechanism for cytokinesis in these plant cells. Their paper describes step by step how it unfolds.
|

