| Oct 31, 2014 |
Cell division, minus the cells
|
|
(Nanowerk News) The process of cell division is central to life. The last stage, when two daughter cells split from each other, has fascinated scientists since the dawn of cell biology in the Victorian era. For just as long, it has been notoriously difficult to study this final step, when the dividing cell creates a furrow before cleaving in two.
|
|
The name given to this process by those early biologists, cytokinesis, translates as "cell movement" and captures the sense of a highly active and organized series of events. Scientists have now learned much more about the proteins involved and their behavior, and yet a central mystery remains: How does the cell signal where the furrow should be?
|
|
There is a simple reason for that blind spot: It’s hard to see and test such a complex feat in living cells. Now Harvard Medical School systems biologists report in Science ("Spatial organization of cytokinesis signaling reconstituted in a cell-free system") that they have reconstituted cytokinesis—complete with signals that direct molecular traffic—without the cell. Combining frog-egg extracts with lipid membranes that mimic the membrane of the cell, they built a cell-free system that recapitulates how the cleavage furrow is assembled.
|
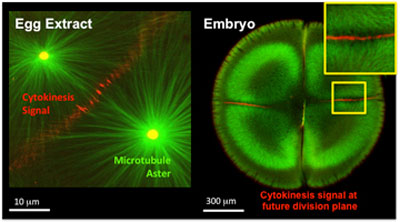 |
| Both the cell-free egg extract system and embryos recruit critical signaling molecules necessary for cytokinesis signaling. In these micrographs, fluorescently labeled microtubules are green and Aurora B, the cytokinesis signal, is red. (Images: Mitchison Lab)
|
|
This cell-free system has two huge advantages: It expands the scale of the furrow-building events, making them easier to see, and it gives the researchers an easy way to manipulate the proteins involved. Quickly removing and returning proteins to see how changes in the molecular players affect cytokinesis is impossible when the cell is whole, but easy when the cellular innards are spread out on a microscope slide.
|
|
The key challenge, though, was that the behavior of cytokinesis is entirely dependent on having a membrane to furrow—and membranes are exactly what have to be removed to make the system cell-free. What made this work possible was the realization that a controlled, flat membrane—made from two layers of artificial lipid supported on glass—could substitute for the curved, constantly moving and complex membrane of the cell.
|
|
“We really built what goes on in the cell,” said Timothy Mitchison, the Hasib Sabbagh Professor of Systems Biology at HMS. “We believe our work is a step forward, not only for understanding the assembly of the cleavage furrow but also, more generally, for understanding the biophysics of cytokinesis signaling.”
|
|
The team includes co-senior authors Mitchison and Christine Field, HMS instructor in systems biology, and co-lead authors Phuong Nguyen, a graduate student in chemical biology, and Aaron Groen, a research fellow in systems biology.
|
|
Their work began with gently crushing unfertilized egg cells from the Xenopus frog and isolating their internal contents. Scientists have previously used such egg extracts to reconstitute microtubule-based structures called mitotic spindles that align chromosomes properly in preparation for cell division. The HMS team went further.
|
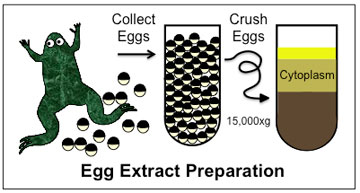 |
| African clawed frog (Xenopus laevis) egg-extract preparation involves collecting unfertilized eggs, crushing them, and separating out fractions of the cytoplasm through centrifugation, a process that divides membranes, organelles and cytoplasm by density. Image: Mitchison Lab African clawed frog (Xenopus laevis) egg-extract preparation involves collecting unfertilized eggs, crushing them, and separating out fractions of the cytoplasm through centrifugation, a process that divides membranes, organelles and cytoplasm by density. (Image: Mitchison Lab)
|
|
To start, the scientists turned to their molecular stockpile to come up with such building blocks as artificial centrosomes and fluorescently labeled microtubules. They then mimicked fertilization and added labeled antibodies or proteins to visualize the self-organization of structures required for the cell division process, using fluorescence microscopy in ways that aren’t possible with actual living cells in tissue cultures.
|
|
The final component was the model of the cell membrane.
|
|
“To really prove that we reconstituted the cytokinesis signal, we needed to add the bilayer membrane and then see if it could recruit the proteins that would be on the cortex of the cell,” Field said about the specialized layer beneath the cell membrane. “That’s the signal to the membrane we were looking for.”
|
|
The scientists saw this signal in part because Field chose to include actin in their system, a cellular component that is often discarded because it makes it so difficult to do experiments with the egg extract. Actin's job in the cell is to assemble into long filaments and meshworks, like Lego blocks fitting together.
|
|
The filaments provide a scaffold that allows other cellular components, including membranes, to move and change shape. So Field reasoned that, despite the difficulty of using actin, it would be important to include it in a system intended to recapitulate the signals involved in membrane furrowing.
|
|
“It’s not only microtubules signaling to the plasma membrane, but also microtubules signaling to the actin cortex that forms on top of the plasma membrane,” Nguyen said. “This phenomenon can be seen only when actin is around.”
|
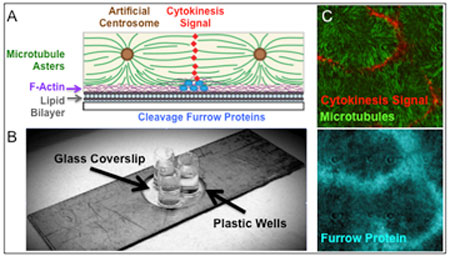 |
| The structural components of the cell-free system that reconstitutes cytokinesis signaling (A-B). Signaling occurs from the midzone, a region of overlapping microtubules between asters, to a specific region in the lipid bilayer where cytokinesis is to occur (C). The lipid bilayers, supported on coverslips, are imaged by Total Internal Fluorescence Microscopy which illuminates a very thin layer of the sample (<200nm), maximizing the signal-to-noise ratio for precise measurements of microtubule dynamics. Sample micrographs are shown on the right. Images: Mitchison LabThe structural components of the cell-free system that reconstitutes cytokinesis signaling (A-B). Signaling occurs from the midzone, a region of overlapping microtubules between asters, to a specific region in the lipid bilayer where cytokinesis is to occur (C). The lipid bilayers, supported on coverslips, are imaged by Total Internal Fluorescence Microscopy which illuminates a very thin layer of the sample (<200nm), maximizing the signal-to-noise ratio for precise measurements of microtubule dynamics. Sample micrographs are shown on the right. (Images: Mitchison Lab)
|
|
There are multiple signaling complexes at work in the cleavage furrow, “talking” from the microtubules to the actin cortex and the cell membrane.
|
|
”The beauty of this system is that we have reconstructed cytokinesis from its individual structural components: actin, microtubules and membranes,” said Groen. “And we can now begin to spatially manipulate these components, which is not possible in live cells. I’m very excited by the possibilities.”
|
|
Their work is science at its most basic, Mitchison said, but cell division is central to embryonic development, stem cell renewal and cancer.
|
|
“These processes we talk about are the same as what's going on in our bone marrow to make new blood cells, for example,” he said. “So clearly understanding this biology is relevant to many types of diseases.”
|



