| Dec 17, 2014 |
Time, energy and storage capacity determine the measurement accuracy of the 'cell computer'
|
|
(Nanowerk News) Researchers from FOM institute AMOLF have discovered what determines the accuracy with which cells can measure chemical concentrations.
|
|
They described the results of their research in two publications: on 24 November online in the Proceedings of the National Academy of Sciences ("Optimal resource allocation in cellular sensing systems") and on 16 December in Physical Review Letters ("Energy Dissipation and Noise Correlations in Biochemical Sensing").
|
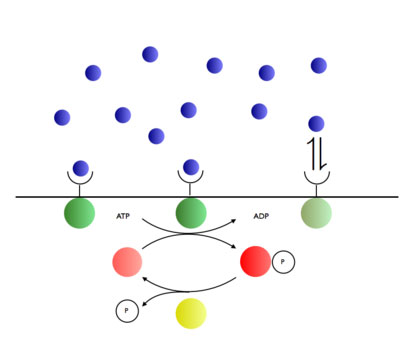 |
| Living cells measure concentrations of molecules (blue) via biochemical networks consisting of proteins that react with each other. Researchers from AMOLF have now developed a theory that describes how the measurement accuracy is determined by the number of proteins, the measurement time and the energy that the system consumes.
|
|
Whether it is bacteria searching for food or human cells that are differentiating into a brain cell, all living cells must respond to signals from their environment. These signals consist of chemical concentrations of nearby molecules.
|
|
Cell computer
|
|
Cells are able to measure these chemical concentrations with unprecedented precision thanks to receptor proteins on their surface, which bind to the surrounding molecules. A network of chemical reactions subsequently transmits the signal into the cell.
|
|
These networks are the information processing machines of a living cell, comparable with a computer. Just like a computer, the cellular system needs time, energy and storage capacity to function. A large difference between a computer and a cellular measurement system is that cellular processes are very noisy, as a result of which it was not yet clear how cells can measure concentrations with such a high precision.
|
|
The AMOLF researchers have now developed a theory that describes the accuracy of a concentration measurement based on the number of proteins present, the time and the energy that the cell invests in each measurement.
|
|
Optimal design
|
|
The researchers first looked at passive cellular detection systems, which consume no energy. In these systems the amount of receptor proteins, the proteins that bind the surrounding molecules, limits the number of measurements the cell can perform. The cell computer takes an average of this number to determine the overall molecule concentration.
|
|
To obtain a more accurate estimate of the concentration, the cellular system must remember measurements from the past. However, this requires time, energy and other proteins to store the information. To the researchers' surprise, these three resources determine the accuracy of the measurements like links in a chain. The precision is limited by the resource that is the least present.
|
|
This observation led the researchers to a design principle not previously identified for this type of system: in an optimal measurement system all of the links are equally strong. The network that enables the bacterium E. coli to find food was found to obey this principle.
|
|
The researchers expect that this design logic of cellular systems can also be used to develop efficient synthetic systems and materials.
|

