| Mar 03, 2015 |
Sizing up cells: Study finds possible regulator of growth
|
|
(Nanowerk News) Modern biology has attained deep knowledge of how cells work, but the mechanisms by which cellular structures assemble and grow to the right size largely remain a mystery. Now, Princeton University researchers may have found the key in a dynamic agglomeration of molecules inside cells.
|
|
This structure is called the nucleolus, a part of the cell typically found under a microscope in groups of small black specks, like poppy seeds in a muffin. The primary function of the nucleolus is to make ribosomes, molecular machines that are crucial to cell growth. The Princeton research team asked a simple question about the nucleolus: do these ribosome factories get bigger as cells that contain them get bigger?
|
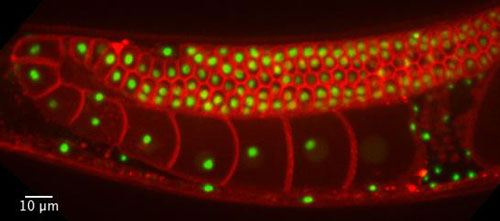 |
| These are C. Elegans cells used in the research. (Image: Brangwynne and Weber)
|
|
"We thought: does the nucleolus' assembly and function depend on the size of the cell?" said Clifford Brangwynne, the lead researcher and an assistant professor of chemical and biological engineering at Princeton. "If this were true, then it could provide a feedback mechanism for regulating cell growth."
|
|
The limit of cellular growth and how cells "choose" their eventual size have long been open questions for biologists. Cells with a common function - muscular, neural, etc. - form a uniform size, but how they do so has been unknown. The mechanism by which cells perform this function, and ways in which it goes wrong, also play an important role in many types of disease, including various forms of cancer.
|
|
"It's well known that the nucleolus drives cell growth," Brangwynne said. "Is it possible that there are simple biophysical rules that directly link the nucleolus and its functions to cell size - allowing the nucleolus to 'know' the size of a cell."
|
|
The nucleolus is one of a number of assemblies inside a cell called organelles; these structures function like organs do in the human body. More specifically, the nucleolus is known as an RNA/protein body - a class of organelle that has no defined boundary that separates it from the surrounding liquid protoplasm. In earlier research, Brangwynne discovered that these structures behave like viscous liquid droplets -- like a drop of oil in water. Brangwynne's work on several RNA/protein bodies suggests that these liquid-like structures assemble through a phase transition -- the same type of mechanism that causes dew drops to condense on blade of grass.
|
|
The group's earlier work led them to consider whether a higher concentration of cellular matter would more easily lead to the phase transition that assembles nucleoli. If so, that could regulate cell size: a small cell with a high concentration of matter would easily assemble nucleoli, but once a cell grew too large, spreading out its material, nucleoli would disassemble and the cell would stop growing.
|
|
"It's like making rock candy from a sugar solution," Brangwynne said. "If you dilute the solution too much, the candy won't grow in the solution."
|
|
Brangwynne and Stephanie Weber, a post-doctoral researcher in the lab, tested the hypothesis through a series of experiments using embryos of a type of worm called a nematode. In an article published online Feb. 19 in the journal Current Biology ("Inverse Size Scaling of the Nucleolus by a Concentration-Dependent Phase Transition"), the researchers detail how they used a technique called RNA interference (RNAi), to increase or decrease the size of the cells, and then examined the effect on the assembly of nucleoli.
|
|
"We took advantage of some useful features of the worm that allowed us to change the concentration of all of the nucleolar components by just changing cell size", Weber said.
|
|
The team was able to derive a mathematical model based on the physics of phase transitions, which explain this size variation. In the model, they show that the assembly and size of nucleoli depend on two factors: the first is the concentration of cellular material (which is related to cell size) the second is the energy of interactions between the molecules in the cell, which determines the saturating concentration for the phase transition. The model explains why nucleoli do not assemble in some cells, and why they grow to larger or smaller sizes in other cells.
|
|
Support for the project was provided in part by the National Institutes of Health, the Searle Scholars Program, the National Science Foundation, and a Damon Runyon Postdoctoral Fellowship.
|
|
Brangwynne said the conclusion raises the possibility that phase transition-driven assembly of the nucleolus limits cell growth -- when the cell is relatively small, nucleoli easily assemble and produce enough ribosomes for cell growth; but when the cell grows larger, it becomes harder to produce enough ribosomes and cell growth tapers.
|
|
Brangwynne is working to directly test this idea. "We've developed some exciting new technology that allows us to precisely measure the dynamics of worm growth, and we're now using it to test whether the rate at which nucleoli produce ribosomes is directly impacted by the biophysical link to cell size that we've uncovered."
|

